The global healthcare landscape is undergoing a profound transformation, driven by rapid innovation in biologics, vaccines, and advanced therapeutics.
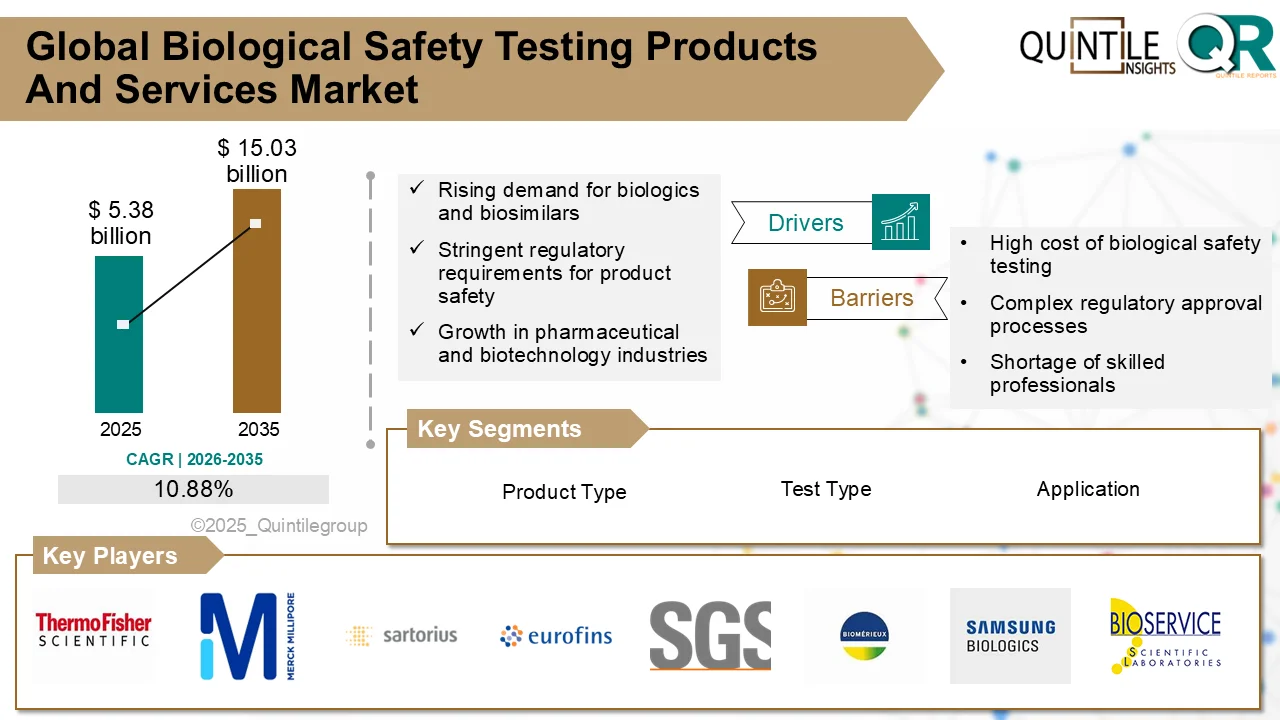
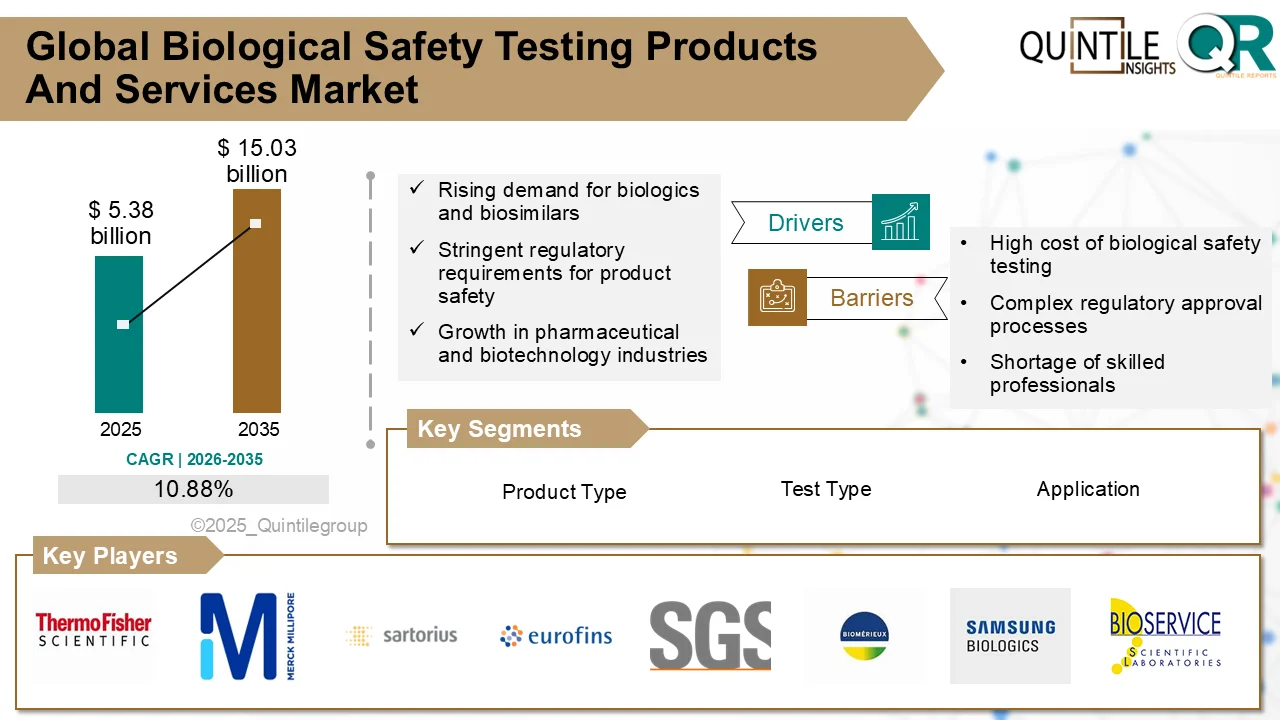
At the center of this transformation lies a critical enabler—biological safety testing products and services. According to the latest industry analysis, the global Biological Safety Testing Products and Services Market is projected to grow from USD 5.38 billion in 2025 to USD 15.03 billion by 2035, registering a strong CAGR of 10.88% over the forecast period.
This remarkable growth reflects the increasing importance of ensuring the safety, purity, and compliance of modern therapeutics. As biologics continue to dominate pharmaceutical pipelines, biological safety testing is no longer optional—it is a strategic necessity.
Request Sample PDF Report: https://www.quintilereports.com/request-sample/1214-biological-safety-testing-products-and-services-market/
Understanding the Biological Safety Testing Ecosystem
Biological safety testing encompasses a wide range of products, technologies, and outsourced services aimed at verifying the safety and integrity of biologics. These include:
- Sterility testing
- Endotoxin detection
- Viral clearance validation
- Adventitious agent detection
- Bioburden analysis
- Cell line authentication
The market serves pharmaceutical companies, biotech firms, contract research organizations (CROs), and contract development and manufacturing organizations (CDMOs).
With biologics accounting for over 35% of the global drug development pipeline, the complexity and volume of safety testing have increased significantly.
Special Discount: https://www.quintilereports.com/request-discount/1214-biological-safety-testing-products-and-services-market/
Key Drivers Fueling Market Growth
1. Regulatory Stringency Across Global Markets
Regulatory bodies worldwide have intensified safety requirements for biologics. Authorities such as:
- U.S. Food and Drug Administration
- European Medicines Agency
- Pharmaceuticals and Medical Devices Agency
mandate multi-layered testing protocols before product approval. These stringent guidelines ensure product safety but also drive demand for advanced testing solutions.
2. Surge in Biologics and Advanced Therapies
The rise of cell and gene therapies, monoclonal antibodies, and recombinant proteins is a major growth catalyst. These therapies require rigorous testing due to their complexity and sensitivity.
Notably:
- Over 60% of new drug approvals are biologics
- Gene therapy pipelines are growing at over 20% CAGR
- Each biologic batch requires multiple layers of safety validation
This surge directly translates into higher demand for biological safety testing services.
3. Technological Advancements Revolutionizing Testing
The integration of advanced technologies is reshaping the industry:
- Next-generation sequencing (NGS)
- AI-powered data analytics
- High-throughput automation systems
These innovations are reducing testing timelines by up to 30–40%, improving accuracy, and enabling faster regulatory approvals.
Buying Now: https://www.quintilereports.com/request-enquiry/1214-biological-safety-testing-products-and-services-market/
4. Rising Cost of Biologics Failure
A single contaminated batch of biologics can lead to losses exceeding USD 500 million. This has shifted safety testing from a compliance requirement to a strategic investment.
Companies now prioritize proactive testing to avoid financial, reputational, and regulatory risks.
Market Challenges and Restraints
Despite strong growth potential, the market faces several challenges:
High Testing Costs
Biological safety testing can account for 15–25% of total drug development costs, making it expensive for small biotech firms.
Regulatory Fragmentation
Differences in requirements across regions increase complexity and delay global launches.
Talent Shortage
The industry faces a shortage of skilled professionals, with demand exceeding supply by 20–30% in key markets.
Market Saturation in Developed Regions
North America and Western Europe are highly competitive, limiting growth opportunities for new entrants.
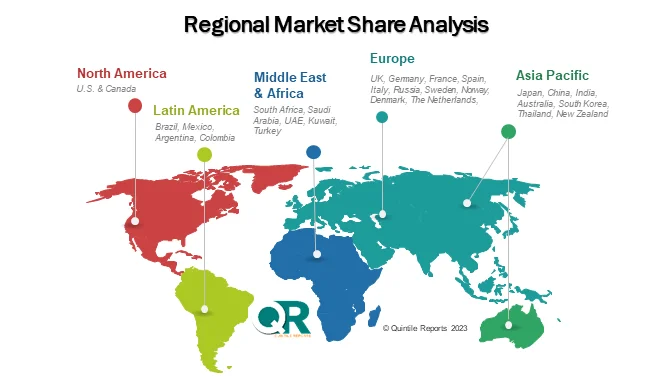
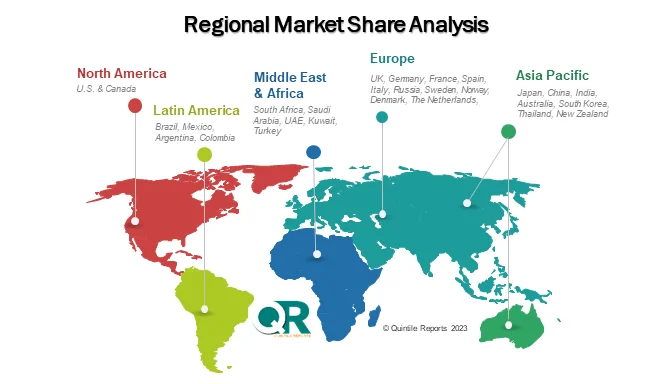
Regional Insights
North America – Market Leader
North America dominates the global market, accounting for over 40% of total revenue. The region benefits from:
- Strong regulatory frameworks
- High R&D investment
- Rapid adoption of advanced technologies
The United States, in particular, leads due to its large biopharmaceutical ecosystem and extensive clinical trial activity.
Europe – Innovation Meets Regulation
Europe remains a key market, driven by:
- Strict regulatory compliance
- Strong biosimilar pipeline
- Advanced research infrastructure
Countries like Germany and Switzerland are at the forefront of biosafety innovation.
Asia Pacific – Fastest Growing Region
Asia Pacific is witnessing the fastest growth, fueled by:
- Expansion of biologics manufacturing
- Increasing clinical trials
- Government support for biotech
China and India are emerging as major hubs for outsourced testing services.
Emerging Markets
Regions like Latin America and the Middle East & Africa are gradually expanding, supported by improving healthcare infrastructure and regulatory alignment.
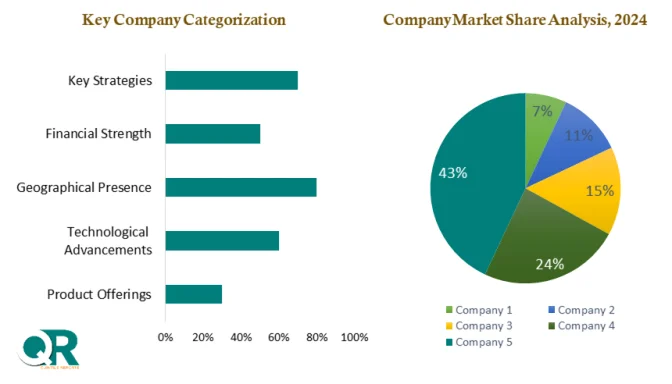
Competitive Landscape
The biological safety testing market is highly competitive and expertise-driven. Key players differentiate themselves through:
- Regulatory compliance expertise
- End-to-end service offerings
- Advanced testing technologies
- Strategic partnerships
Leading companies include:
- Thermo Fisher Scientific
- Merck KGaA
- Sartorius AG
- Eurofins Scientific
- Lonza Group
- WuXi AppTec
These companies are investing heavily in automation, AI integration, and global expansion to maintain their competitive edge.
Recent Developments
- In April 2025, Eurofins expanded its biosafety services in the U.S. with new sterilization solutions.
- In May 2025, advanced gene therapy trials highlighted increasing demand for biosafety testing.
These developments underscore the growing importance of safety testing in next-generation therapeutics.
Future Outlook: A Decade of Opportunity
Looking ahead, the biological safety testing market is poised for sustained growth. Key trends shaping the future include:
- Increased outsourcing to CROs and CDMOs
- Expansion of personalized medicine
- Integration of AI and machine learning
- Growth of emerging biotech hubs
Companies that prioritize innovation, regulatory expertise, and strategic partnerships will be best positioned to capitalize on this growth.
Conclusion
The Biological Safety Testing Products and Services Market is entering a transformative phase. As biologics continue to reshape modern medicine, the need for reliable, efficient, and compliant safety testing will only intensify.
From regulatory mandates to technological advancements, multiple factors are converging to drive market expansion. While challenges such as cost and talent shortages remain, the long-term outlook is overwhelmingly positive.
For stakeholders across the pharmaceutical and biotechnology ecosystem, biological safety testing is not just a requirement—it is a cornerstone of future success.
FAQs for Key Opinion Leaders (KOLs)
1. Why is biological safety testing becoming critical in modern drug development?
Biological safety testing ensures the safety, purity, and efficacy of biologics, which are complex and highly sensitive therapies requiring strict validation.
2. What technologies are transforming biological safety testing?
Technologies such as next-generation sequencing (NGS), AI-driven analytics, and automation are improving efficiency, accuracy, and turnaround times.
3. Which regions offer the highest growth potential?
Asia Pacific is the fastest-growing region due to expanding biologics manufacturing and increasing clinical trials.
4. What are the biggest challenges in the market?
High testing costs, regulatory complexity, and a shortage of skilled professionals are the key challenges.
5. How are CROs and CDMOs influencing the market?
They are enabling cost-effective, scalable testing solutions and helping companies meet regulatory requirements efficiently.
6. What role does AI play in biological safety testing?
AI enhances data analysis, reduces testing time, and improves detection accuracy for contaminants and anomalies.
7. How can companies stay competitive in this market?
By investing in advanced technologies, forming strategic partnerships, and ensuring regulatory compliance across regions.
Our Latest Publication:
Biological Safety Testing Products and Services Market (2026 – 2035)
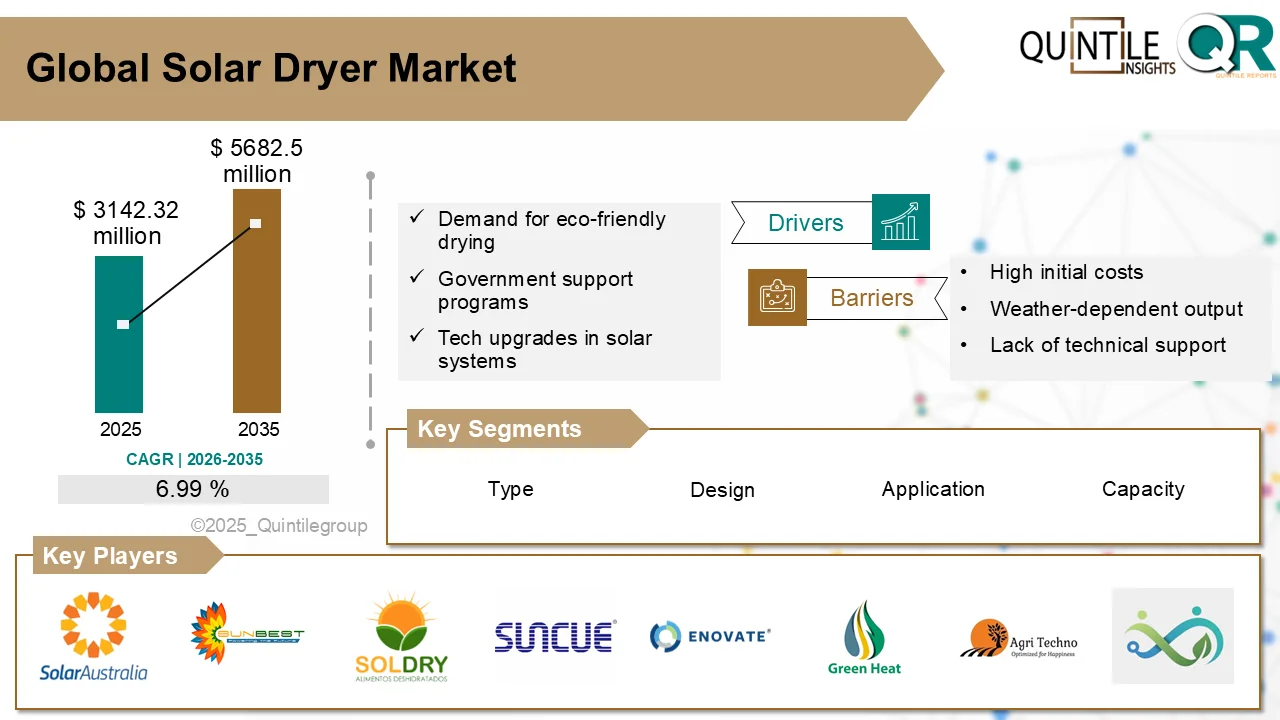
Solar Dryer Market Set for Sustainable Growth as Global Food Systems Transform
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.