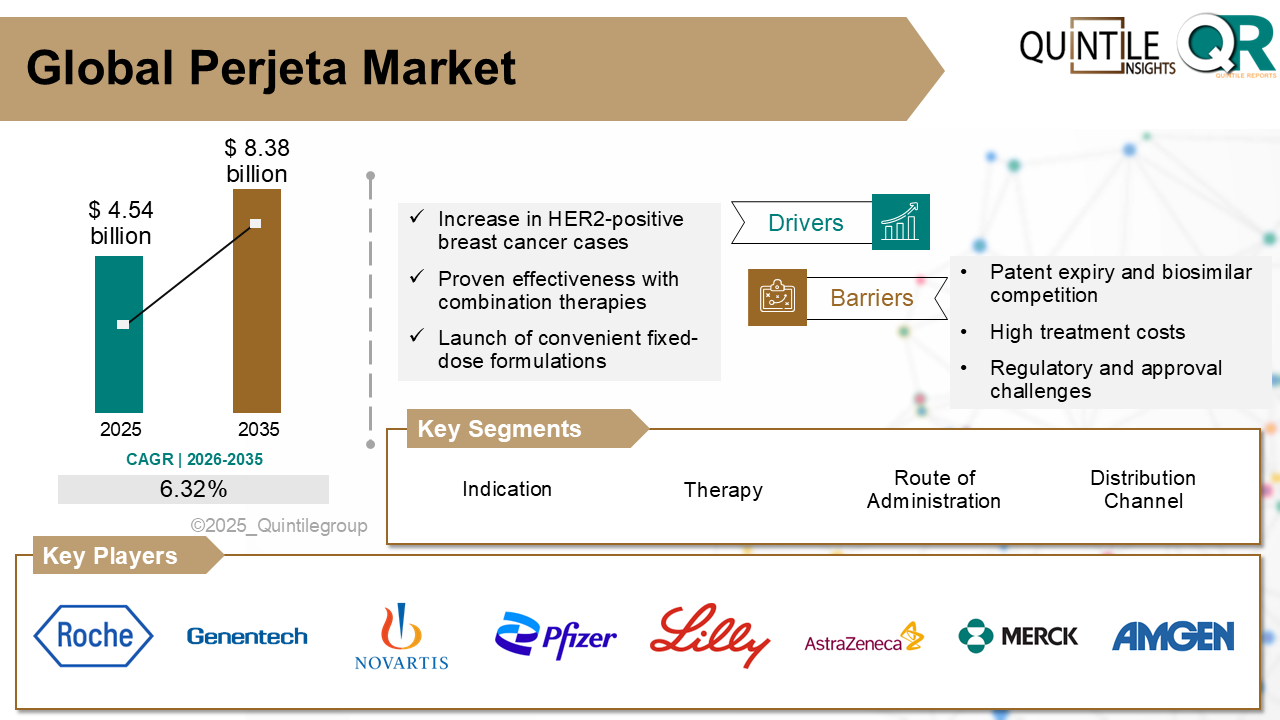
The global Perjeta market is poised for steady expansion over the next decade, driven by rising incidence of HER2-positive breast cancer, continued clinical validation, and strategic innovation in drug delivery.
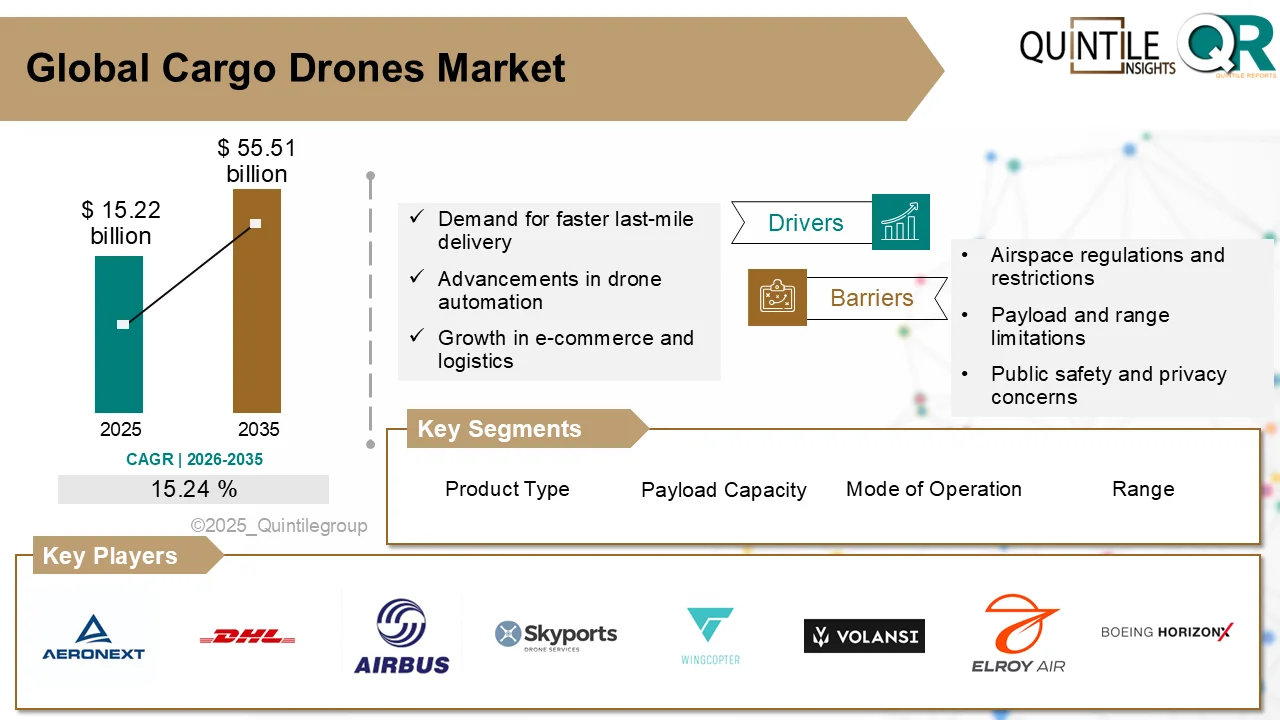
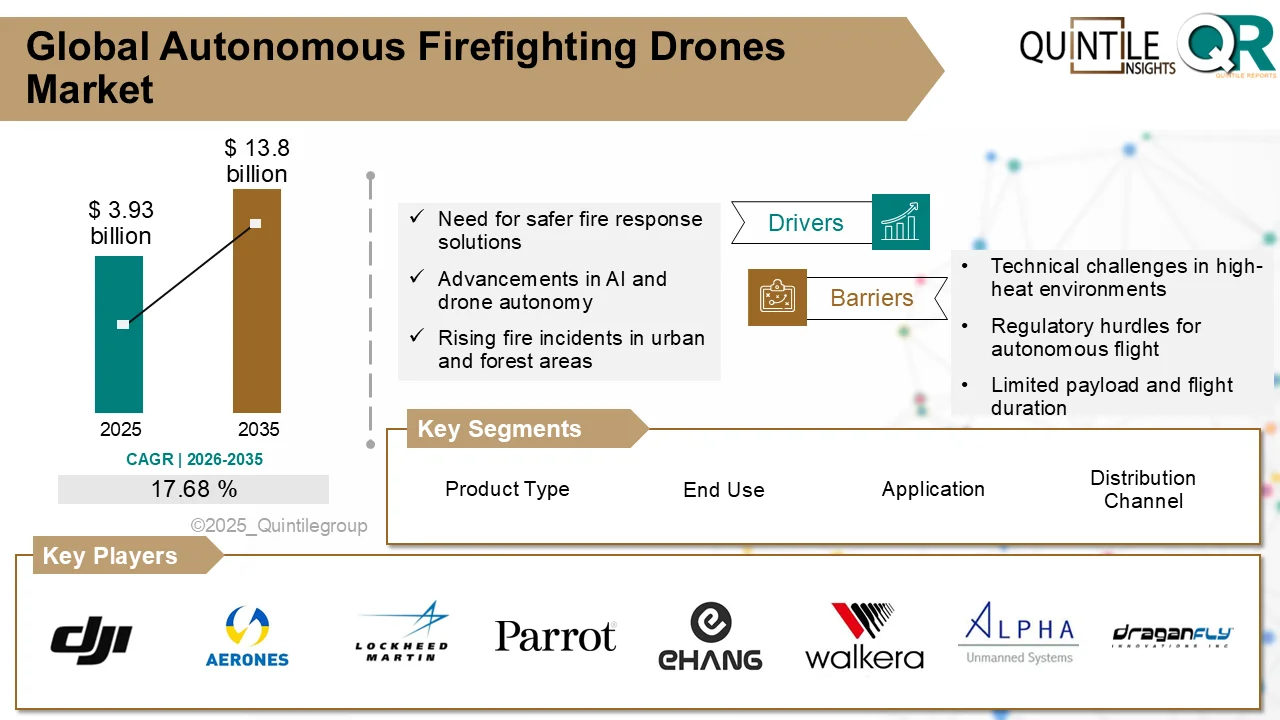
According to the latest analysis by Quintile Research, the market was valued at USD 4.54 billion in 2026 and is projected to reach USD 8.38 billion by 2035, registering a compound annual growth rate (CAGR) of 6.32% during the forecast period.
Perjeta, the brand name for pertuzumab developed by Roche and its U.S. subsidiary Genentech, is a monoclonal antibody targeting HER2-positive breast cancer. It is commonly administered in combination with trastuzumab and chemotherapy for both early-stage and metastatic settings. Over the years, Perjeta has become a cornerstone of dual HER2 blockade therapy, particularly following strong clinical outcomes demonstrated in pivotal trials.
Request Sample PDF Report: https://www.quintilereports.com/request-sample/1108-perjeta-market/
One of the major growth drivers for the Perjeta market is the increasing global prevalence of breast cancer, especially HER2-positive subtypes, which account for approximately 15–20% of all diagnosed cases. Improved screening initiatives, earlier diagnosis, and expanding access to targeted oncology therapies have significantly enhanced treatment uptake. Clinical studies such as CLEOPATRA and APHINITY have reinforced the therapeutic value of Perjeta-based regimens, showing meaningful improvements in survival rates and reductions in disease progression.
Innovation in formulation has further strengthened the product’s competitive positioning. The introduction of PHESGO, a fixed-dose subcutaneous combination of pertuzumab and trastuzumab, has simplified administration compared to traditional intravenous infusions. This advancement has reduced treatment times, enhanced patient convenience, and enabled broader adoption in outpatient and homecare settings. As healthcare systems increasingly emphasize efficiency and patient-centered care, such innovations are expected to contribute meaningfully to sustained market growth.
Special Discount: https://www.quintilereports.com/request-discount/1108-perjeta-market/
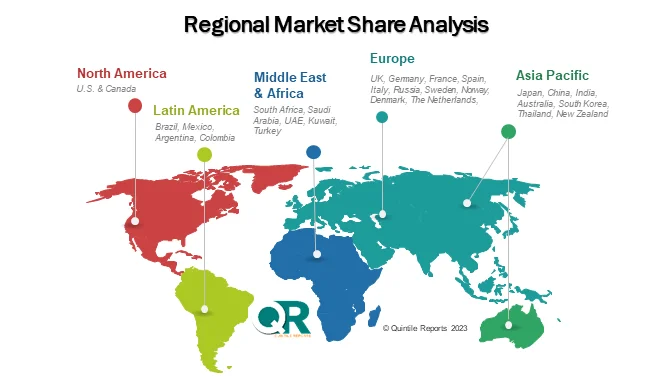
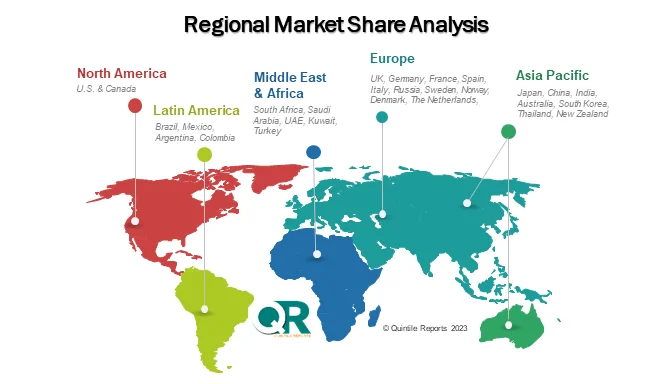
Regionally, North America remains the leading revenue contributor, supported by advanced oncology infrastructure, strong reimbursement frameworks, and widespread HER2 testing. The United States continues to dominate the market, benefiting from guideline endorsements and high biologics adoption rates. Europe follows closely, with countries such as Germany, the United Kingdom, France, and Italy expanding access to targeted breast cancer therapies through evolving reimbursement models and personalized medicine strategies.
Meanwhile, the Asia-Pacific region is emerging as the fastest-growing market. Rapid healthcare modernization, rising cancer awareness, and expanding middle-class healthcare spending in China, Japan, South Korea, and India are accelerating demand. Government-led screening programs and improved oncology infrastructure are also broadening patient access. Latin America and the Middle East & Africa are witnessing gradual but promising growth, supported by public-private partnerships and increasing investments in cancer care services.
Despite its positive outlook, the Perjeta market faces notable challenges. The expiration of key patents in the United States and European Union in 2025 is expected to open the door for biosimilar competition beginning in 2026. Several biosimilar candidates are progressing through regulatory pathways, potentially offering lower-cost alternatives that may pressure pricing and market share, particularly in cost-sensitive regions.
Buying Now: https://www.quintilereports.com/request-enquiry/1108-perjeta-market/
Pricing and reimbursement dynamics represent additional constraints. As a high-cost biologic therapy, Perjeta faces scrutiny from payers and health technology assessment bodies. Although negotiated discounts and value-based pricing agreements have supported continued access in major markets, the anticipated entry of biosimilars could intensify competitive pricing strategies.
Competitive activity within the oncology landscape remains strong. In addition to Roche and Genentech, major pharmaceutical players such as Novartis, Pfizer, AstraZeneca, Merck & Co., and Amgen are actively investing in oncology research, combination regimens, and biologic innovations. Strategic collaborations, label expansions, and data-driven regulatory submissions are shaping the competitive landscape.
Looking ahead, the Perjeta market is expected to maintain its upward trajectory through 2035, supported by ongoing research and development, improved drug delivery technologies, and expanding global access to targeted therapies. While biosimilar entry and pricing pressures may reshape market dynamics, strong clinical evidence and established physician confidence are likely to sustain demand for Perjeta-based regimens in the years to come.
Quintile Research concludes that stakeholders who strategically adapt to evolving regulatory, competitive, and reimbursement environments will be best positioned to capitalize on the long-term growth potential of the global Perjeta market.
1. What is the projected growth rate of the Perjeta market from 2026 to 2035?
The global Perjeta market is expected to grow at a CAGR of 6.32% during the forecast period. The market is projected to increase from USD 4.54 billion in 2026 to USD 8.38 billion by 2035, driven by rising HER2-positive breast cancer cases and expanding targeted therapy adoption.
2. What is Perjeta and who manufactures it?
Perjeta is the brand name for pertuzumab, a monoclonal antibody used to treat HER2-positive breast cancer. It is developed and marketed by Roche and its U.S. biotechnology subsidiary Genentech. It is typically used in combination with trastuzumab and chemotherapy.
3. What are the primary indications covered in the Perjeta market report?
The report segments the market based on the following indications:
-
Breast Cancer
-
HER2-Positive Breast Cancer
-
Metastatic Breast Cancer
-
Early Breast Cancer
HER2-positive breast cancer remains the dominant segment due to strong clinical evidence supporting dual HER2 blockade therapy.
4. What factors are driving the growth of the Perjeta market?
Major growth drivers include:
-
Rising global prevalence of HER2-positive breast cancer
-
Strong clinical trial outcomes (e.g., CLEOPATRA and APHINITY studies)
-
Introduction of subcutaneous fixed-dose combinations (PHESGO)
-
Expanding healthcare infrastructure in emerging markets
-
Increased HER2 testing and early cancer detection programs
5. What are the major challenges facing the Perjeta market?
Key challenges include:
-
Patent expiration in the U.S. and EU in 2025
-
Entry of biosimilars starting in 2026
-
Pricing and reimbursement pressure in cost-sensitive regions
-
Legal disputes over patent protection
-
Slower biosimilar substitution in biologics compared to generics
These factors may impact pricing strategies and market share distribution.
6. Which regions dominate the Perjeta market?
North America currently leads the market due to advanced oncology infrastructure and strong reimbursement systems. Europe follows with steady growth supported by personalized medicine adoption.
Asia-Pacific is the fastest-growing region, driven by increasing breast cancer incidence and expanding healthcare access in countries like China, Japan, South Korea, and India.
7. How will biosimilars impact the Perjeta market?
The expiration of patents is expected to allow biosimilar versions of pertuzumab to enter the market from 2026 onward. These alternatives may offer 20–30% cost savings, potentially affecting Roche’s market share. However, strong brand loyalty, established clinical data, and innovative formulations may help maintain competitive positioning.
8. What are the key companies operating in the Perjeta market ecosystem?
Major companies influencing the competitive landscape include:
These players are actively involved in oncology research, biologics development, and combination therapies.
9. What distribution channels are covered in the report?
The Perjeta market is segmented by distribution channel into:
-
Hospital Pharmacies
-
Retail Pharmacies
-
Online Pharmacies
Hospital pharmacies currently dominate due to intravenous administration requirements.
10. Why should stakeholders invest in this Perjeta market report?
The report provides:
-
Historical analysis (2017–2024)
-
Forecast projections (2026–2035)
-
Detailed segmentation insights
-
Regional and country-level analysis
-
Competitive benchmarking
-
Market dynamics including drivers, restraints, and opportunities
It serves as a strategic decision-making tool for pharmaceutical companies, investors, healthcare providers, and policymakers seeking long-term growth opportunities in the HER2-targeted therapy landscape.
Our Latest Publication
Perjeta Market (2026 – 2035)
Our Latest News:
Feminine Hygiene Products Market to Reach USD 78.73 Million by 2035 Amid Sustainability and Innovation Push
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.