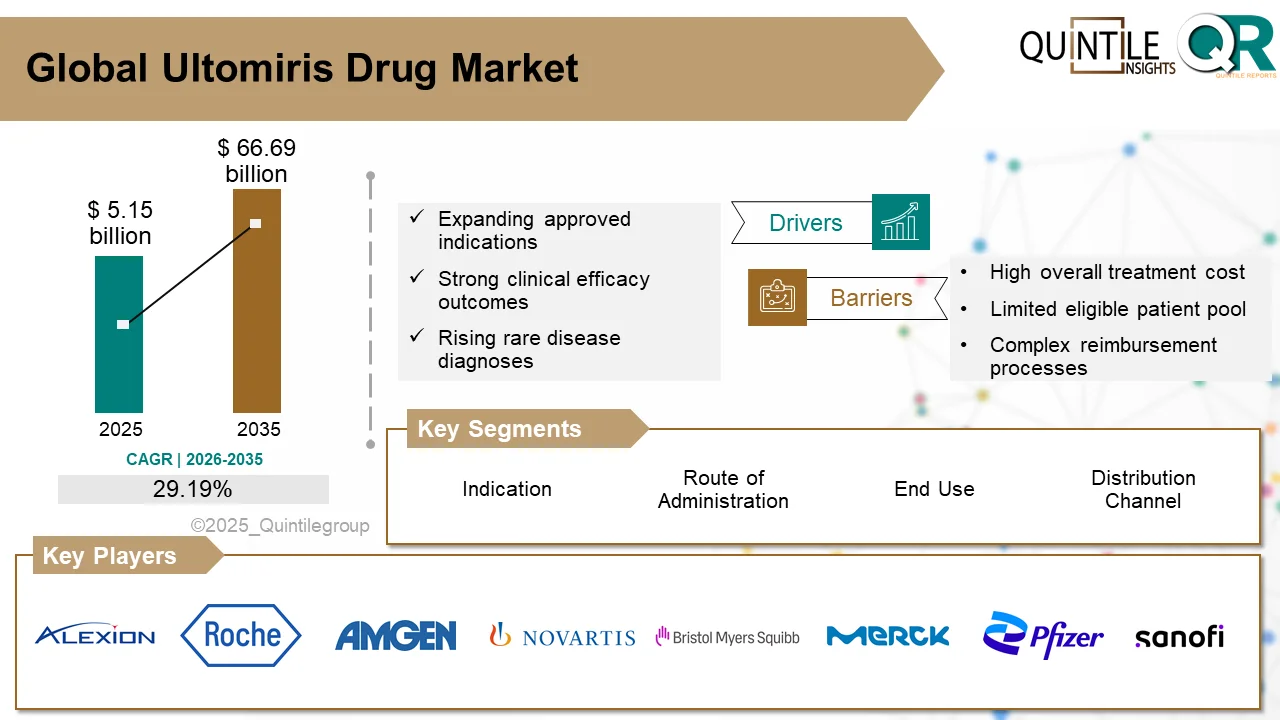
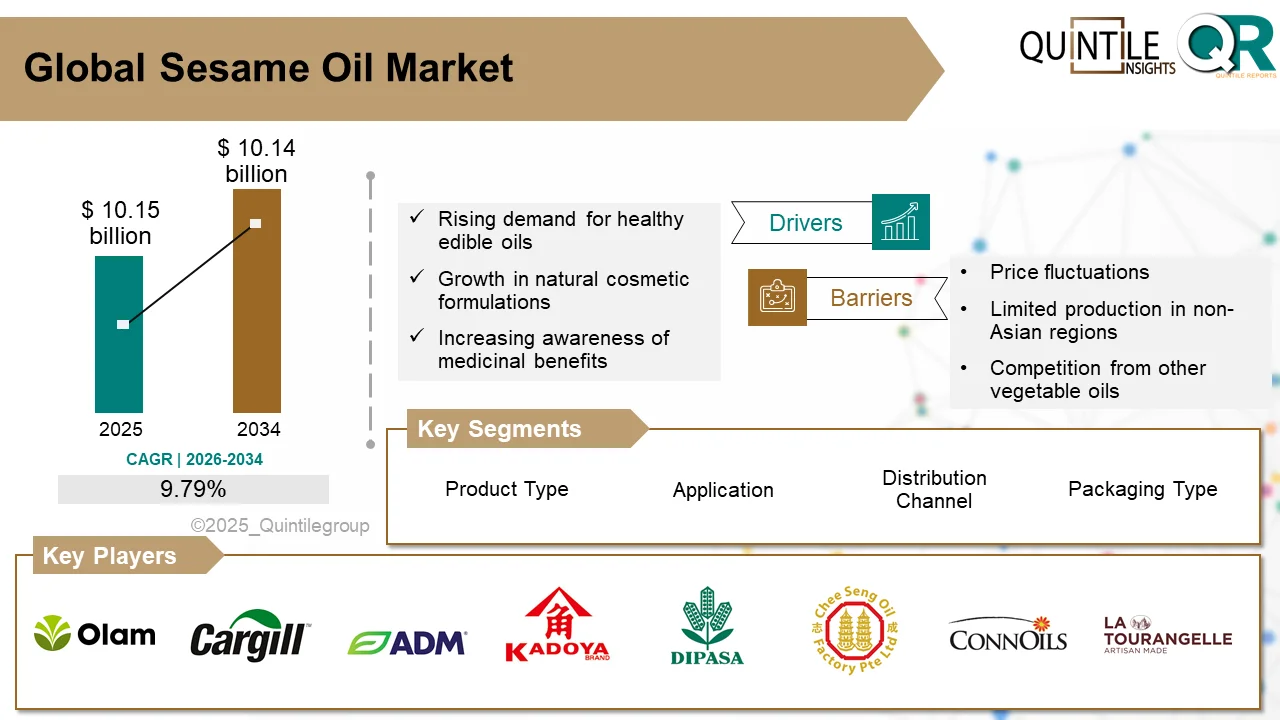
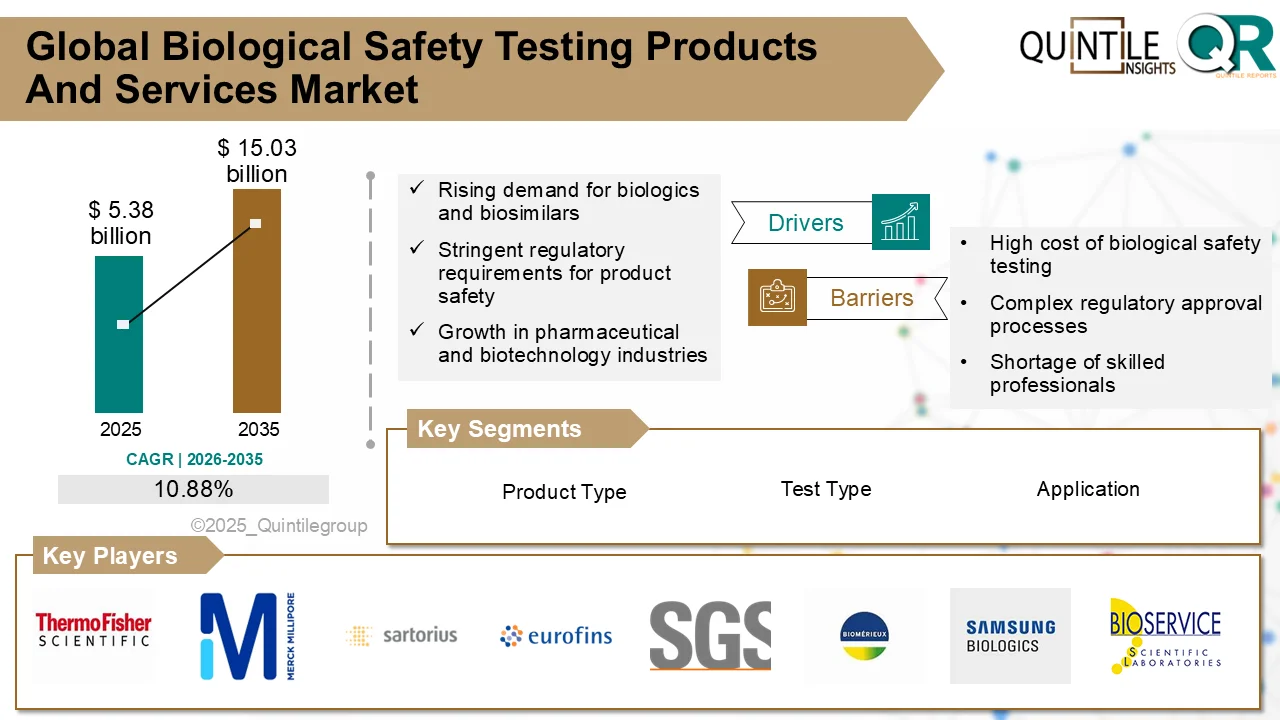
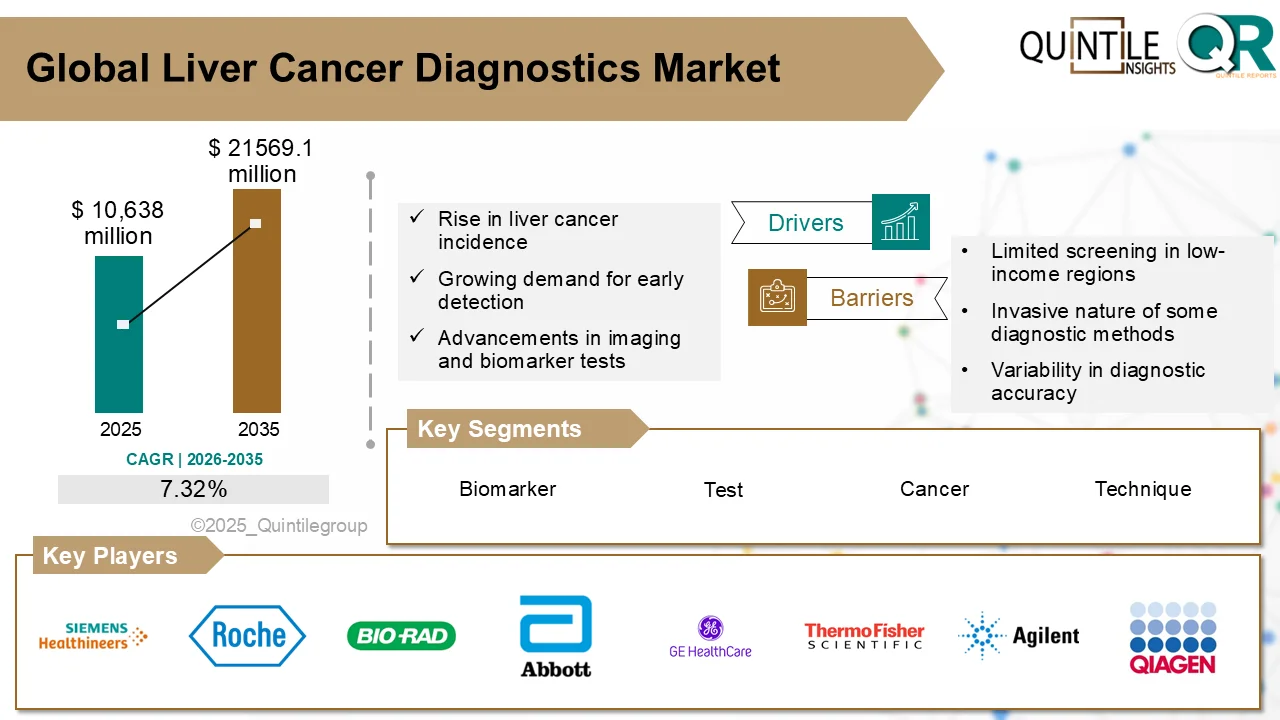
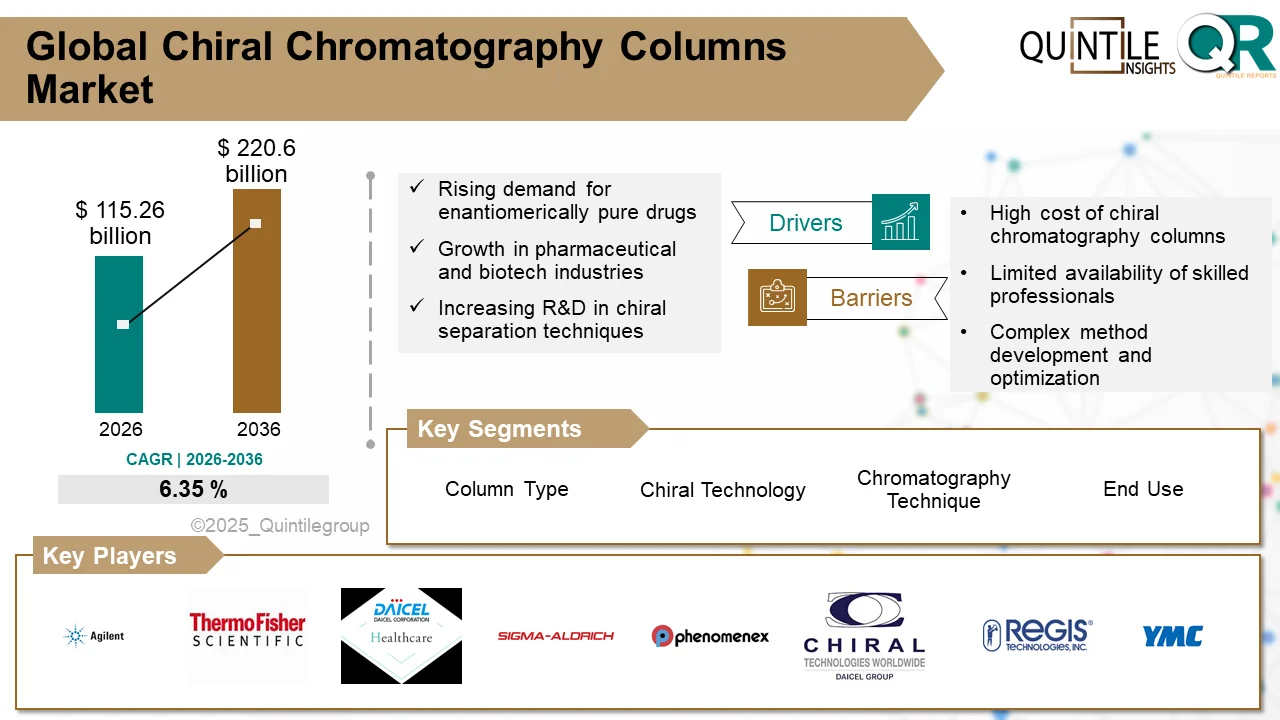
The global Ultomiris Drug Market is entering a transformative phase as healthcare systems worldwide increase investments in rare disease therapies, biologics, and advanced immunology treatments.
According to recent industry analysis, the market was valued at USD 5.15 billion in 2026 and is projected to reach USD 66.69 billion by 2035, expanding at an impressive CAGR of 29.19% during the forecast period.
The remarkable growth trajectory of the Ultomiris Drug Market reflects the rising importance of complement inhibitor therapies in modern medicine. Ultomiris (ravulizumab), developed as a long-acting complement C5 inhibitor, has rapidly become a preferred treatment option for several rare autoimmune and hematologic disorders, including paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (aHUS), generalized myasthenia gravis (gMG), and neuromyelitis optica spectrum disorder (NMOSD).
As pharmaceutical companies continue to focus on targeted biologics and precision therapies, the Ultomiris Drug Market is gaining momentum across major healthcare economies including the United States, Germany, Japan, and emerging Asia-Pacific markets.
Request Sample PDF Report: https://www.quintilereports.com/request-sample/1094-ultomiris-drug-market/
Rising Burden of Rare Diseases Fuels Market Demand
One of the primary growth drivers supporting the expansion of the Ultomiris Drug Market is the increasing diagnosis of complement-mediated rare diseases. Improvements in genomic diagnostics, specialist referral systems, and awareness campaigns are helping healthcare providers identify patients earlier than ever before.
Rare disorders such as PNH and aHUS were historically underdiagnosed due to their complex clinical presentation. However, modern diagnostic tools and enhanced physician education are significantly improving detection rates. As a result, demand for advanced therapies like Ultomiris continues to surge.
Healthcare experts note that patients increasingly prefer therapies that reduce treatment frequency while maintaining efficacy. Ultomiris offers an important advantage over earlier therapies because it requires dosing every eight weeks instead of biweekly infusions associated with older complement inhibitors.
Special Discount: https://www.quintilereports.com/request-discount/1094-ultomiris-drug-market/
This reduced treatment burden is positively influencing physician adoption and patient compliance across the Ultomiris Drug Market.
Expanded Regulatory Approvals Strengthen Commercial Potential
Another major catalyst accelerating the Ultomiris Drug Market is the continuous expansion of regulatory approvals across multiple indications.
The therapy has already secured approvals for PNH, aHUS, gMG, and NMOSD in key global markets. In March 2024, the U.S. FDA approved Ultomiris for anti-AQP4 antibody-positive NMOSD, making it the first long-acting C5 inhibitor for this condition.
Clinical evidence from the CHAMPION-NMOSD Phase III trial demonstrated a 98.6% reduction in relapse risk, reinforcing physician confidence and further strengthening the position of the Ultomiris Drug Market.
Similarly, Health Canada approved Ultomiris for NMOSD treatment in January 2023, highlighting the expanding international acceptance of complement inhibitor therapies.
Pharmaceutical analysts believe that additional label expansions and pediatric approvals could further accelerate revenue generation opportunities over the next decade.
Buying Now: https://www.quintilereports.com/request-enquiry/1094-ultomiris-drug-market/
North America Maintains Market Leadership
North America currently dominates the Ultomiris Drug Market, supported by advanced healthcare infrastructure, favorable reimbursement frameworks, and high awareness regarding rare diseases.
The United States represents the largest revenue-generating region due to strong FDA support, extensive specialist networks, and broad insurance coverage for orphan drugs. Hospitals and specialty clinics across the country are increasingly adopting long-acting biologics to improve patient convenience and reduce healthcare utilization costs.
The presence of major biopharmaceutical innovators and ongoing clinical trials continues to strengthen the regional outlook of the Ultomiris Drug Market.
In addition, patient advocacy organizations and rare disease awareness campaigns are improving access to advanced biologics, further driving commercial expansion.
Europe Focuses on Rare Disease Infrastructure
Europe represents another significant contributor to the Ultomiris Drug Market, particularly in countries such as Germany, France, and the United Kingdom.
European healthcare systems are emphasizing structured rare disease treatment protocols and centralized reimbursement systems. Germany remains one of the strongest markets in Europe due to efficient diagnostic pathways and strong physician adoption of complement inhibitors.
Despite strict pricing negotiations and health technology assessments, the region continues to witness stable demand growth. Healthcare providers increasingly recognize the long-term value of therapies that reduce relapse rates and hospitalizations.
As the European Medicines Agency continues evaluating newer indications and biologic innovations, the Ultomiris Drug Market is expected to maintain strong momentum throughout the forecast period.
Asia-Pacific Emerges as the Fastest-Growing Region
Asia-Pacific is rapidly becoming the fastest-growing regional segment within the Ultomiris Drug Market.
Countries such as Japan, South Korea, China, and Australia are investing heavily in advanced biologics and specialty healthcare infrastructure. Japan, in particular, has emerged as a major growth center due to early regulatory approvals and comprehensive orphan drug reimbursement programs.
Improving diagnostic capabilities and rising awareness of autoimmune disorders are contributing to stronger therapy adoption across Asia-Pacific healthcare systems.
Medical experts believe that the growing middle-class population and expansion of specialty hospitals will create long-term growth opportunities for the Ultomiris Drug Market in emerging economies.
However, high therapy costs remain a challenge in several lower-income nations where reimbursement support remains limited.
Competitive Landscape Intensifies
The competitive environment within the Ultomiris Drug Market is becoming increasingly dynamic as pharmaceutical companies invest in next-generation complement inhibitors and biosimilar research.
Alexion Pharmaceuticals, a subsidiary of AstraZeneca, currently holds a dominant market position through its strong patent portfolio, physician relationships, and orphan drug exclusivity.
Ultomiris has successfully positioned itself as a strategic successor to Soliris by offering extended dosing intervals and improved patient convenience.
Nevertheless, the market faces growing competition from alternative therapies under development by major biopharmaceutical companies including Roche, Novartis, Sanofi, Pfizer, Amgen, and Regeneron.
Emerging complement inhibitors with alternative mechanisms of action may intensify competition in the coming years. In addition, biosimilar entry after patent expiration could reshape pricing structures across the Ultomiris Drug Market.
Despite these competitive pressures, analysts expect continued growth due to strong clinical performance and increasing global demand for rare disease therapies.
High Therapy Costs Remain a Key Challenge
Although the Ultomiris Drug Market demonstrates exceptional growth potential, affordability remains one of the industry’s most pressing concerns.
Biologic therapies for rare diseases often involve extremely high development and manufacturing costs. Consequently, treatment expenses can place significant financial pressure on healthcare systems and patients.
In developing regions, limited reimbursement coverage and restricted access to specialty infusion centers continue to hinder market penetration.
Healthcare policymakers are increasingly evaluating value-based reimbursement models and risk-sharing agreements to improve access to innovative therapies while managing overall healthcare spending.
Industry stakeholders believe that broader insurance coverage and expanded government support programs will play a critical role in the long-term sustainability of the Ultomiris Drug Market.
Innovation and Precision Medicine Shape Future Outlook
The future outlook for the Ultomiris Drug Market remains highly promising as biotechnology innovation accelerates globally.
Advancements in precision medicine, biomarker-based diagnostics, and immunology research are expected to support further expansion of complement-targeted therapies.
Pharmaceutical companies are actively exploring combination therapies, next-generation biologics, and expanded autoimmune applications that may further broaden the commercial potential of Ultomiris.
In addition, growing collaboration between healthcare providers, biotech firms, and patient advocacy groups is strengthening the rare disease ecosystem worldwide.
As healthcare systems increasingly prioritize personalized treatment approaches, the Ultomiris Drug Market is positioned to remain a critical component of the evolving biologics landscape through 2035.
Conclusion
The global Ultomiris Drug Market is witnessing unprecedented expansion driven by rising rare disease prevalence, regulatory approvals, extended dosing benefits, and advances in biologic therapies.
While affordability challenges and competitive pressures remain key concerns, the market’s long-term outlook remains highly optimistic due to growing patient demand and continuous innovation in complement inhibitor treatments.
Industry participants that focus on accessibility, clinical expansion, and strategic partnerships are likely to gain significant competitive advantages in the evolving Ultomiris Drug Market over the next decade.
FAQs for Key Opinion Leaders (KOLs)
Q1. What is driving the rapid growth of the Ultomiris Drug Market?
The market is primarily driven by increasing diagnosis of rare autoimmune diseases, expanded regulatory approvals, improved patient adherence, and growing adoption of long-acting biologics.
Q2. Which diseases are currently treated using Ultomiris?
Ultomiris is approved for PNH, aHUS, generalized myasthenia gravis (gMG), and neuromyelitis optica spectrum disorder (NMOSD).
Q3. Why is Ultomiris preferred over older complement inhibitors?
Ultomiris requires dosing every eight weeks, significantly reducing treatment burden compared to older therapies requiring biweekly infusions.
Q4. Which region dominates the Ultomiris Drug Market?
North America currently leads the market due to advanced healthcare systems, strong reimbursement support, and early regulatory approvals.
Q5. What challenges could impact future market growth?
High therapy costs, reimbursement limitations, regulatory scrutiny, and emerging biosimilar competition are major market challenges.
Q6. Which companies are competing in the Ultomiris Drug Market?
Major players include Alexion Pharmaceuticals (AstraZeneca), Roche, Novartis, Pfizer, Sanofi, Amgen, Takeda, Regeneron, and Bristol-Myers Squibb.
Q7. How is precision medicine influencing the Ultomiris Drug Market?
Precision medicine is improving diagnosis, patient stratification, and targeted treatment approaches, increasing adoption of complement inhibitor therapies.
Our Latest Publication:
Ultomiris Drug Market (2026 – 2035)
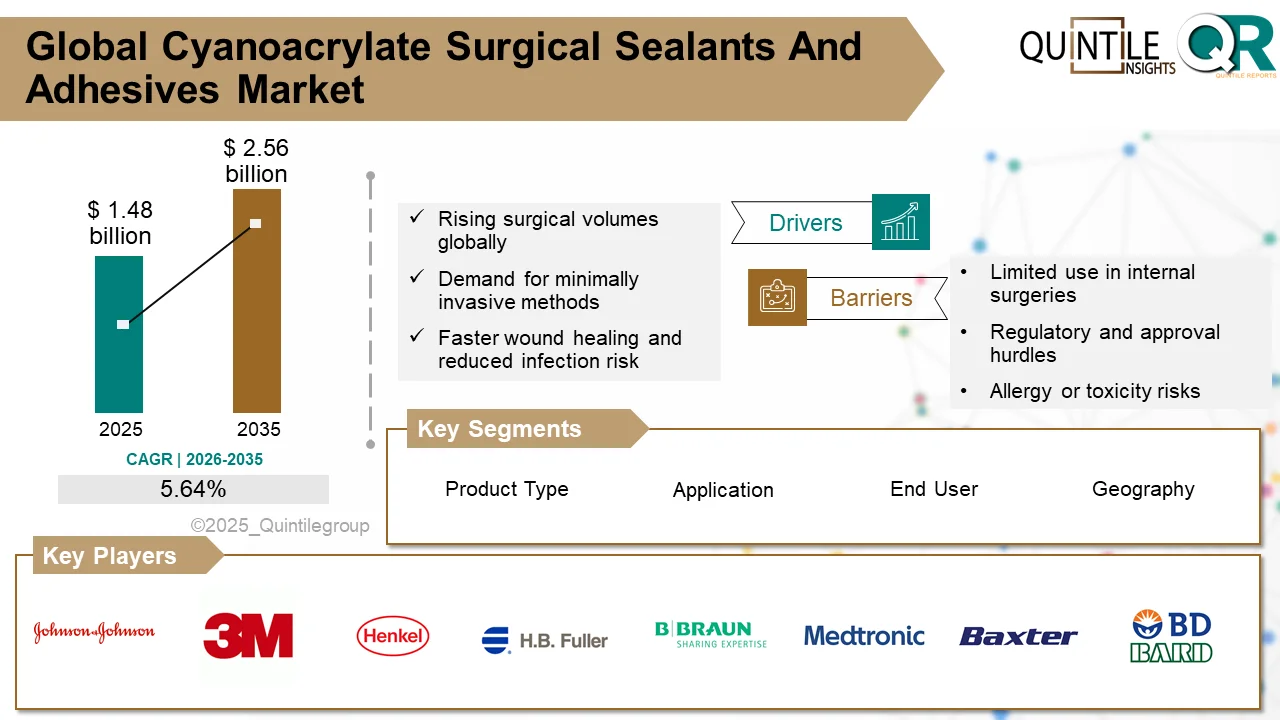
Cyanoacrylate Surgical Sealants Market to Reach USD 2.56 Billion by 2035 Globally
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.