The global Respiratory Syncytial Virus (RSV) Diagnostics Market is projected to experience steady growth over the next decade as healthcare systems place increasing emphasis on early detection and prevention of respiratory infections.
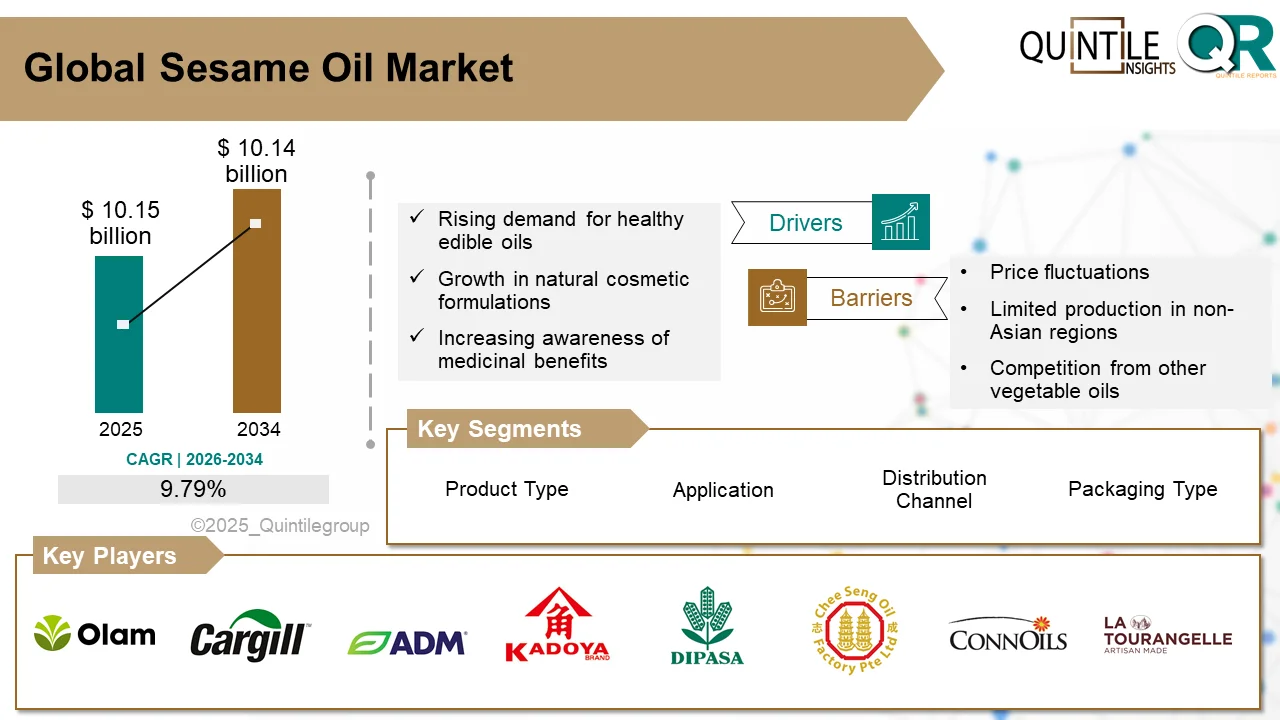
According to recent industry analysis, the market was valued at USD 7.01 billion in 2025 and is expected to reach USD 9.45 billion by 2034, expanding at a compound annual growth rate (CAGR) of 6.28% between 2025 and 2034.
Respiratory Syncytial Virus is a leading cause of lower respiratory tract infections, particularly among infants, elderly individuals, and people with weakened immune systems. As the prevalence of RSV infections continues to rise globally, the demand for reliable and rapid diagnostic solutions is increasing. Accurate detection plays a critical role in enabling timely treatment, preventing complications, and limiting the spread of infection during seasonal outbreaks.
Request Sample PDF Report: https://www.quintilereports.com/request-sample/1032-respiratory-syncytial-virus-diagnostics-market/
Healthcare experts highlight that RSV contributes significantly to global pediatric hospitalizations. The World Health Organization reports that the virus is responsible for millions of hospital admissions and a large number of deaths each year among children under five. This growing clinical burden has intensified efforts by governments, healthcare institutions, and diagnostic companies to strengthen RSV testing capabilities worldwide.
Technological advancements are playing a major role in transforming the RSV diagnostics landscape. One of the most widely used methods is reverse transcription polymerase chain reaction (RT-PCR), which is considered the gold standard for RSV detection due to its high sensitivity and accuracy. Molecular diagnostic technologies like PCR allow clinicians to detect viral genetic material quickly and reliably, improving patient management and clinical outcomes.
Special Discount: https://www.quintilereports.com/request-discount/1032-respiratory-syncytial-virus-diagnostics-market/
In addition to molecular testing, rapid diagnostic tests (RDTs) and point-of-care (POC) diagnostic kits are gaining widespread adoption. These tools are particularly valuable in outpatient clinics, emergency departments, and healthcare settings with limited laboratory infrastructure. Rapid tests enable healthcare professionals to obtain results in a short time, allowing quicker clinical decision-making and more efficient treatment planning.
Another emerging trend in the market is the adoption of multiplex diagnostic panels, which can simultaneously detect RSV along with other respiratory pathogens such as influenza and COVID-19. These comprehensive panels improve diagnostic efficiency and reduce the time required for identifying the specific cause of respiratory symptoms.
Growing awareness about the clinical impact of RSV is also supporting market growth. Pediatric and geriatric healthcare providers are increasingly recommending routine screening for respiratory viruses during peak infection seasons. In many countries, public health authorities are strengthening surveillance systems to monitor RSV activity and improve outbreak preparedness.
Buying Now: https://www.quintilereports.com/request-enquiry/1032-respiratory-syncytial-virus-diagnostics-market/
Government initiatives and funding programs are further supporting the expansion of diagnostic infrastructure. Many healthcare systems are integrating RSV testing into national disease monitoring programs, helping improve data collection and early detection strategies.
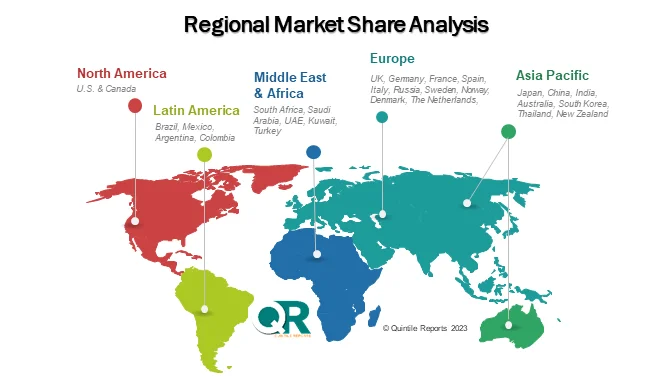
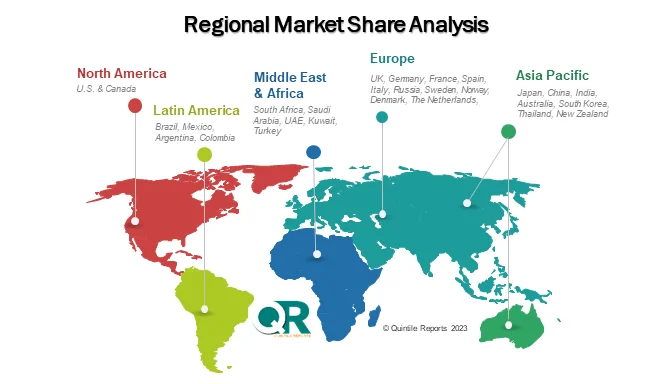
Regionally, North America holds a dominant share of the RSV diagnostics market due to its advanced healthcare infrastructure, strong diagnostic technology adoption, and active disease surveillance programs. Organizations such as the Centers for Disease Control and Prevention play an important role in monitoring RSV trends and supporting research initiatives.
Europe also represents a significant market, with countries including the United Kingdom, Germany, and France investing in improved diagnostic capabilities and respiratory disease awareness programs.
Meanwhile, the Asia-Pacific region is expected to witness the fastest growth during the forecast period. Rising healthcare investments, increasing RSV incidence, and expanding access to molecular diagnostics in countries such as India, China, and Japan are driving demand across the region.
Key industry players are focusing on innovation and strategic partnerships to strengthen their market position. Companies are developing cost-effective diagnostic solutions, expanding respiratory testing panels, and investing in emerging markets to increase accessibility.
Major companies operating in the RSV diagnostics market include Abbott Laboratories, Roche Diagnostics, Thermo Fisher Scientific, Bio-Rad Laboratories, Hologic Inc., Siemens Healthineers, QIAGEN N.V., and Becton, Dickinson and Company, among others.
Overall, the Respiratory Syncytial Virus Diagnostics Market is expected to maintain strong growth momentum through 2034, supported by technological innovation, increasing healthcare awareness, and rising demand for early disease detection solutions worldwide.
1. What is the Respiratory Syncytial Virus Diagnostics Market?
The Respiratory Syncytial Virus Diagnostics Market refers to the global industry focused on diagnostic technologies used to detect RSV infections. The market includes rapid tests, molecular diagnostics, antigen detection tests, and other advanced solutions designed to identify Respiratory Syncytial Virus infections quickly and accurately.
2. What is the expected growth of the Respiratory Syncytial Virus Diagnostics Market?
The Respiratory Syncytial Virus Diagnostics Market was valued at USD 7.01 billion in 2025 and is projected to reach USD 9.45 billion by 2034, expanding at a CAGR of 6.28% during the forecast period. The increasing demand for early diagnosis and improved healthcare infrastructure are driving the growth of the Respiratory Syncytial Virus Diagnostics Market worldwide.
3. What factors are driving the Respiratory Syncytial Virus Diagnostics Market?
The growth of the Respiratory Syncytial Virus Diagnostics Market is driven by the rising incidence of RSV infections, technological advancements in molecular diagnostics, growing awareness about respiratory diseases, and increasing demand for rapid diagnostic testing. Healthcare organizations are also investing more in RSV surveillance programs, further supporting the market expansion.
4. Which technologies are commonly used in the Respiratory Syncytial Virus Diagnostics Market?
Technologies used in the Respiratory Syncytial Virus Diagnostics Market include PCR-based molecular diagnostics, rapid diagnostic tests (RDTs), antigen detection tests, and immunoassay-based testing systems. Among these, PCR-based tests are considered the gold standard due to their high sensitivity and accuracy.
5. Who are the major end users in the Respiratory Syncytial Virus Diagnostics Market?
Key end users in the Respiratory Syncytial Virus Diagnostics Market include hospitals, diagnostic laboratories, point-of-care testing centers, research institutes, and home healthcare settings. These facilities rely on advanced diagnostic technologies to detect RSV infections quickly and manage respiratory illnesses effectively.
6. Which regions dominate the Respiratory Syncytial Virus Diagnostics Market?
North America currently dominates the Respiratory Syncytial Virus Diagnostics Market, primarily due to advanced healthcare infrastructure, strong research capabilities, and high adoption of innovative diagnostic technologies. Europe also represents a significant share of the market.
7. Which region is expected to grow the fastest in the Respiratory Syncytial Virus Diagnostics Market?
The Asia-Pacific region is expected to witness the fastest growth in the Respiratory Syncytial Virus Diagnostics Market due to increasing healthcare investments, rising RSV infection rates, and expanding diagnostic capabilities in countries like India, China, and Japan.
8. Who are the key companies in the Respiratory Syncytial Virus Diagnostics Market?
Leading companies operating in the Respiratory Syncytial Virus Diagnostics Market include Abbott Laboratories, Roche Diagnostics, Thermo Fisher Scientific, Bio-Rad Laboratories, Siemens Healthineers, Hologic Inc., and QIAGEN N.V. These companies are focusing on developing innovative diagnostic technologies to strengthen their market position.
Our Latest Publication:
Respiratory Syncytial Virus Diagnostics Market (2026 – 2035)
Large Molecule Drug Discovery Outsourcing Market Set to Reach USD 7.81 Billion by 2035 Amid Rising Biologics Research
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.