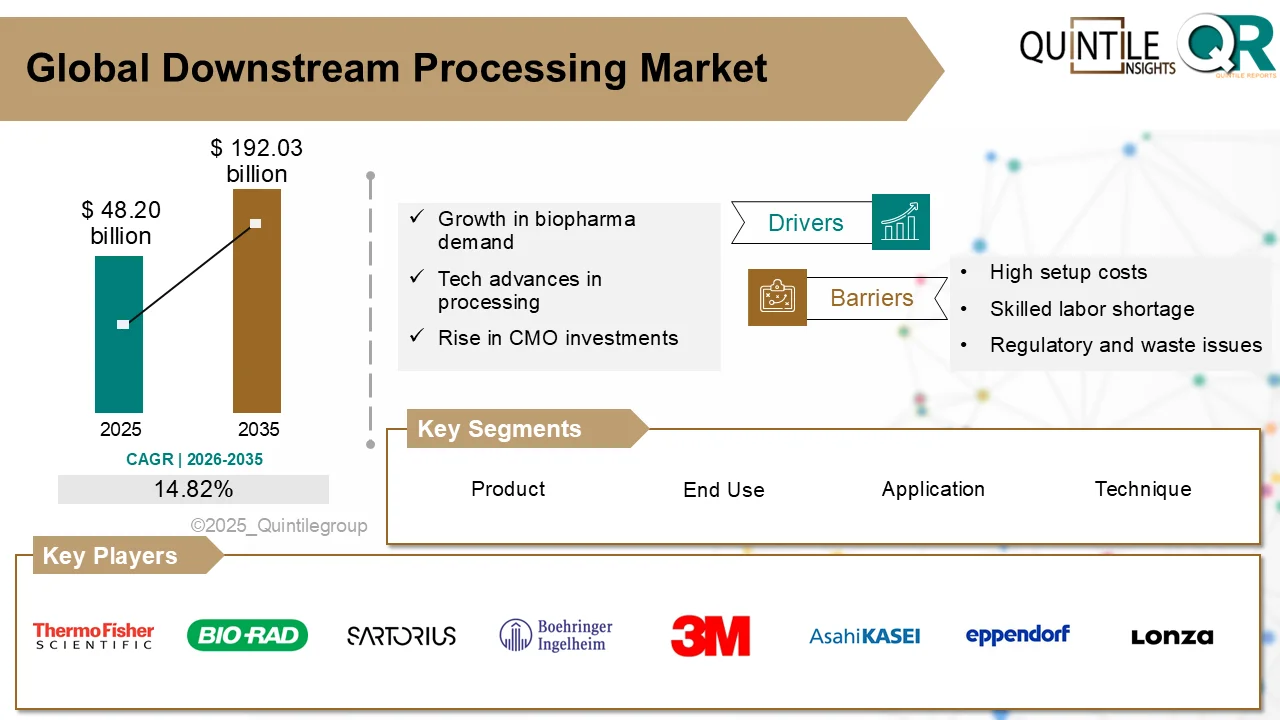
The global Virus Filtration Market is poised for substantial expansion over the next decade, driven by the rapid growth of the biopharmaceutical industry and increasing regulatory focus on viral safety.
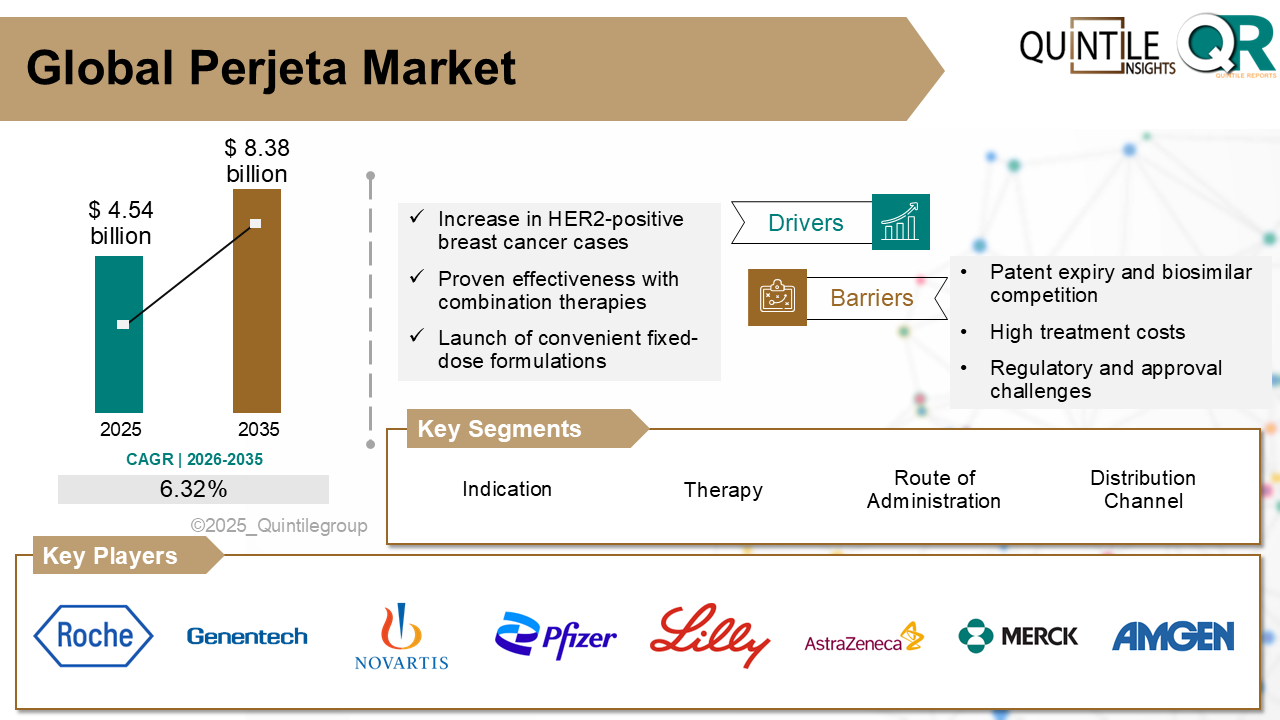
Valued at USD 6.12 billion in 2026, the market is projected to reach USD 22.59 billion by 2035, registering a strong CAGR of 13.91% during the forecast period.
Virus filtration has become a critical component in ensuring the safety, purity, and regulatory compliance of biologics, vaccines, gene therapies, monoclonal antibodies, and blood-derived products. As pharmaceutical and biotechnology manufacturing scales globally, advanced filtration technologies are playing a central role in protecting patient safety and maintaining product integrity.
Request Sample PDF Report: https://www.quintilereports.com/request-sample/1088-virus-filtration-market/
Modern virus filtration solutions incorporate membrane-based systems, nanofiltration, depth filtration, and chromatography techniques. These technologies are increasingly supported by automation, AI-driven monitoring tools, and single-use systems that enhance operational efficiency while reducing contamination risks.
Growth Drivers Accelerating Market Expansion
One of the primary growth catalysts is the rising demand for biologics and advanced therapies. The increasing prevalence of chronic diseases and the global push toward personalized medicine have amplified the production of complex biologic drugs that require stringent viral clearance validation.
Regulatory authorities such as the U.S. Food and Drug Administration and the European Medicines Agency continue to enforce rigorous viral safety standards. These mandates compel manufacturers to adopt validated, high-performance virus filtration systems to meet compliance requirements.
Technological innovation is also reshaping the competitive landscape. High-throughput membranes, improved nanotechnology filters, and AI-enabled process optimization tools are increasing viral removal efficiency while minimizing product loss. In addition, the adoption of disposable and single-use filtration systems is gaining momentum, especially among emerging biotech companies seeking cost-effective and scalable manufacturing solutions.
Asia-Pacific is emerging as the fastest-growing regional market, supported by government investments in pharmaceutical infrastructure and the rapid expansion of biologics manufacturing in countries such as China and India.
𝐒𝐏𝐄𝐂𝐈𝐀𝐋 𝐃𝐈𝐒𝐂𝐎𝐔𝐍𝐓: https://www.quintilereports.com/request-discount/1088-virus-filtration-market/
Competitive Landscape and Strategic Developments
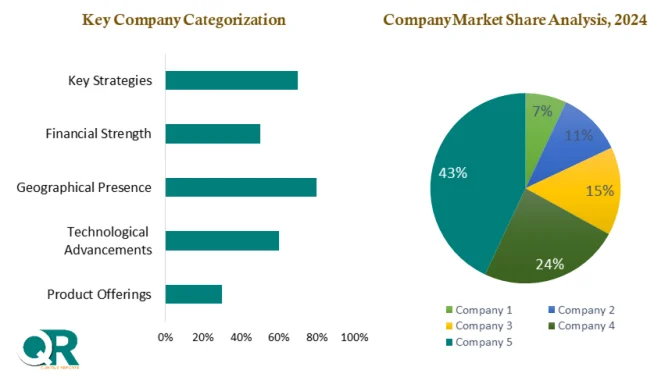
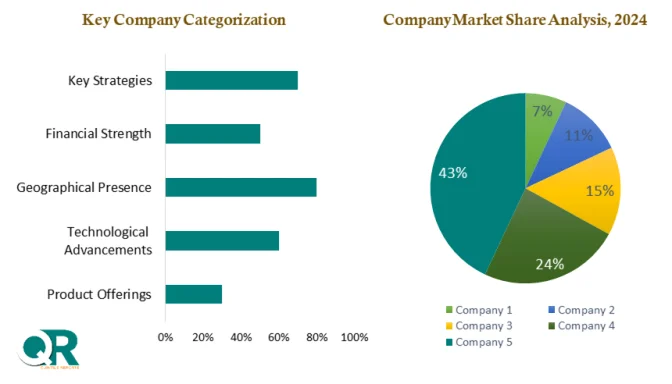
The market remains highly competitive, with global players focusing on innovation, portfolio expansion, and strategic acquisitions. Companies such as Merck KGaA, Thermo Fisher Scientific, Sartorius AG, and Danaher Corporation are strengthening their virus filtration offerings through research investments and partnerships.
In February 2025, Thermo Fisher Scientific announced an agreement to acquire Solventum’s Purification & Filtration business, enhancing its capabilities in advanced filtration solutions for biopharmaceutical manufacturers. Such strategic initiatives reflect the industry’s ongoing consolidation and commitment to technology leadership.
Additionally, innovative companies like Charles River Laboratories and Lonza Group are integrating comprehensive validation services and regulatory support into their offerings, creating value-added solutions for clients navigating complex approval pathways.
Buying Now: https://www.quintilereports.com/request-enquiry/1088-virus-filtration-market/
Challenges and Market Considerations
Despite strong growth momentum, the virus filtration market faces certain constraints. High capital costs associated with advanced filtration equipment can pose challenges, particularly for smaller manufacturers. Moreover, the validation and regulatory approval processes are complex and time-intensive, increasing operational burdens.
Supply chain disruptions—especially shortages of specialized membranes and reagents—have highlighted vulnerabilities within global manufacturing networks. Companies are responding by diversifying supplier bases and investing in regional production capabilities to enhance resilience.
Outlook Through 2035
Looking ahead, the virus filtration market is expected to remain on a robust growth trajectory. The expansion of gene therapies, mRNA vaccines, and biosimilars will continue to fuel demand for high-efficiency viral clearance technologies. Automation, digital integration, and sustainability-focused innovations will further redefine operational standards across bioprocessing facilities worldwide.
As global healthcare systems emphasize safety, compliance, and quality assurance, virus filtration will remain a foundational technology supporting next-generation biologics manufacturing.
With sustained regulatory backing, technological innovation, and rising biopharma output, the global virus filtration market stands positioned for transformative growth through 2035.
1. What is driving the growth of the Virus Filtration Market?
The market is primarily driven by the rising production of biologics such as vaccines, monoclonal antibodies, and gene therapies. Regulatory mandates from agencies like the U.S. Food and Drug Administration and the European Medicines Agency requiring strict viral safety validation are also accelerating adoption. Technological advancements in high-throughput membranes and AI-based monitoring further support market expansion.
2. What is the projected market size by 2035?
The global Virus Filtration Market is projected to reach USD 22.59 billion by 2035, growing at a CAGR of 13.91% from 2026 to 2035.
3. Why is virus filtration critical in biopharmaceutical manufacturing?
Virus filtration ensures the removal of viral contaminants during biologics production. It protects patient safety, ensures regulatory compliance, and maintains the integrity of high-value therapies such as monoclonal antibodies, blood products, and advanced cell and gene therapies.
4. Which technologies are commonly used in virus filtration?
Common technologies include membrane-based filtration, nanofiltration, chromatography, and depth filtration systems. Modern solutions increasingly integrate automation and AI-enabled monitoring tools to enhance performance and reduce contamination risks.
5. Which regions are leading the Virus Filtration Market?
North America leads due to a strong biopharmaceutical ecosystem and strict regulatory standards. Europe follows closely with robust R&D investments. Asia-Pacific is the fastest-growing region, driven by expanding pharmaceutical manufacturing capabilities in countries like China and India.
6. Who are the key players in the market?
Major companies operating in the Virus Filtration Market include:
-
Thermo Fisher Scientific
-
Sartorius AG
-
Danaher Corporation
-
Charles River Laboratories
These companies focus on innovation, regulatory compliance, and strategic acquisitions to strengthen their global presence.
7. What challenges does the market face?
Key challenges include high capital costs of advanced filtration systems, complex regulatory validation processes, and supply chain disruptions related to specialized membranes and reagents.
8. How is technology transforming virus filtration?
Automation, AI-powered monitoring, single-use systems, and nanotechnology-enhanced membranes are improving viral clearance efficiency, reducing contamination risk, and optimizing production scalability in biopharma manufacturing.
9. What role do single-use systems play in market growth?
Single-use filtration systems reduce cross-contamination risk, lower cleaning validation costs, and provide flexibility for small and mid-sized biotech companies. They are especially popular in emerging markets and fast-scaling production environments.
10. What is the long-term outlook for the Virus Filtration Market?
The market is expected to maintain strong double-digit growth through 2035, fueled by increasing gene therapy approvals, mRNA vaccine development, biosimilars production, and heightened global emphasis on viral safety and quality assurance.
Our Latest Publication
Virus Filtration Market (2026 – 2035)
Our Latest News:
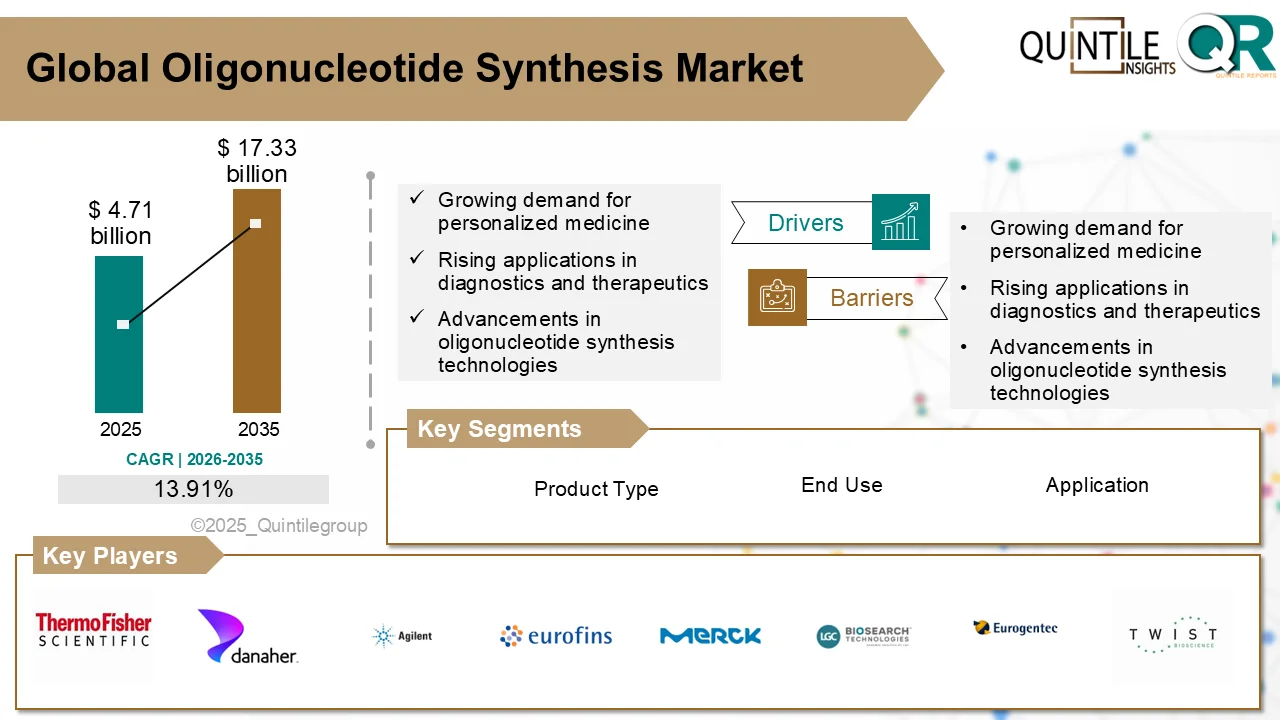
Oligonucleotide Synthesis Market Projected to Reach USD 17.33 Billion by 2035 Amid Precision Medicine Expansion
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.