The global Pharmaceutical Impurity Synthesis and Isolation Services Market is poised for steady expansion, projected to grow from USD 2.14 billion in 2025 to USD 3.86 billion by 2034, at a CAGR of 8.57%.
This growth reflects the increasing importance of impurity profiling in ensuring drug safety, efficacy, and compliance with stringent global regulations.
As pharmaceutical and biotechnology companies continue to develop more complex drug molecules, the need for specialized impurity synthesis and isolation services has intensified. Regulatory authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) are enforcing stricter guidelines on impurity thresholds, pushing companies to invest in advanced analytical solutions.
Request Sample PDF Report: https://www.quintilereports.com/request-sample/1011-pharmaceutical-impurity-synthesis-and-isolation-services-market/
A key driver behind this market growth is the rising complexity of modern therapeutics, including biologics, peptides, and oligonucleotides. These advanced therapies require highly precise impurity identification and characterization to meet safety standards. As a result, contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) are expanding their service portfolios to include comprehensive impurity profiling, synthesis of reference standards, and forced degradation studies.
Technological advancements are also transforming the landscape. Tools such as high-performance liquid chromatography (HPLC), mass spectrometry, and nuclear magnetic resonance (NMR) spectroscopy are enabling more accurate detection and structural elucidation of impurities. These innovations not only improve analytical accuracy but also accelerate drug development timelines.
Outsourcing trends are further fueling market growth. Pharmaceutical companies are increasingly partnering with specialized service providers to reduce operational costs and enhance efficiency. This shift allows drug developers to focus on core competencies while leveraging external expertise for impurity analysis and compliance requirements.
Discount Now: https://www.quintilereports.com/request-discount/1011-pharmaceutical-impurity-synthesis-and-isolation-services-market/
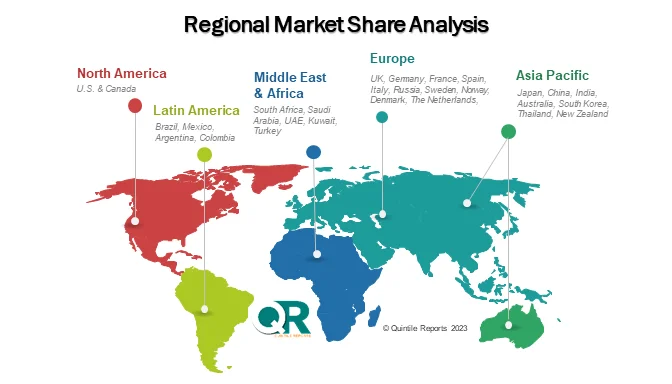
Regionally, North America leads the market due to its strong pharmaceutical base and strict regulatory framework. Europe follows closely, supported by innovation hubs in countries like Germany and the UK. Meanwhile, the Asia-Pacific region—particularly India and China—is emerging as a high-growth area due to cost advantages and expanding pharmaceutical manufacturing capabilities. These regions are becoming preferred destinations for outsourcing impurity-related services.
Despite the positive outlook, the market faces challenges such as regulatory complexities, supply chain disruptions, and the need for highly skilled professionals. However, strategic collaborations, investments in R&D, and expansion into emerging markets are expected to mitigate these challenges and sustain long-term growth.
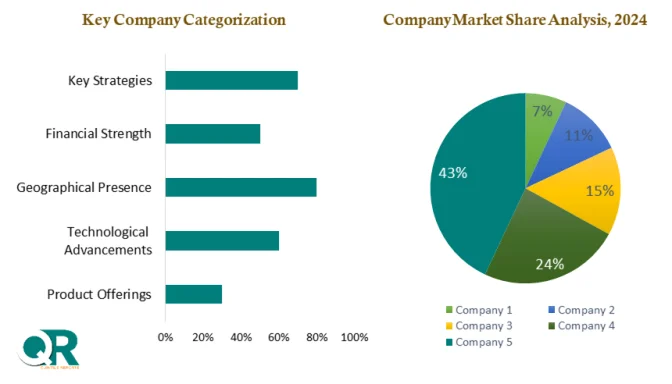
Key players in the market, including leading analytical and life sciences companies, are focusing on innovation, partnerships, and geographic expansion to strengthen their competitive positions. As drug development becomes increasingly sophisticated, the role of impurity synthesis and isolation services will remain critical in ensuring the delivery of safe and effective therapies.
The Pharmaceutical Impurity Synthesis and Isolation Services Market is experiencing significant growth, driven by increasing regulatory scrutiny and the rising complexity of drug formulations. According to industry insights, the Pharmaceutical Impurity Synthesis and Isolation Services Market is projected to grow from USD 2.14 billion in 2025 to USD 3.86 billion by 2034, registering a CAGR of 8.57%.
A major factor contributing to the expansion of the Pharmaceutical Impurity Synthesis and Isolation Services Market is the growing emphasis on drug safety and quality. Regulatory authorities such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) require detailed impurity profiling to ensure pharmaceutical products meet strict safety standards. This has increased the reliance on specialized impurity synthesis and isolation services.
The Pharmaceutical Impurity Synthesis and Isolation Services Market is also benefiting from the rapid advancement of analytical technologies. Techniques such as high-performance liquid chromatography (HPLC), mass spectrometry, and nuclear magnetic resonance (NMR) spectroscopy are enabling more accurate impurity detection and structural characterization. These technological improvements are helping companies reduce development timelines and enhance product quality.
Another key driver of the Pharmaceutical Impurity Synthesis and Isolation Services Market is the rise in outsourcing across the pharmaceutical industry. Companies are increasingly collaborating with contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) to streamline operations and reduce costs. This trend is expected to further accelerate the growth of the Pharmaceutical Impurity Synthesis and Isolation Services Market over the coming years.
The increasing development of complex drug molecules, including biologics, peptides, and oligonucleotides, is further boosting demand within the Pharmaceutical Impurity Synthesis and Isolation Services Market. These advanced therapies require highly specialized impurity analysis, creating new opportunities for service providers to expand their capabilities.
Regionally, North America dominates the Pharmaceutical Impurity Synthesis and Isolation Services Market due to its well-established pharmaceutical infrastructure and strict regulatory environment. Europe follows closely, supported by strong research and development activities. Meanwhile, the Asia-Pacific region is emerging as a key growth hub for the Pharmaceutical Impurity Synthesis and Isolation Services Market, driven by cost advantages and expanding pharmaceutical manufacturing capabilities in countries like India and China.
Despite strong growth prospects, the Pharmaceutical Impurity Synthesis and Isolation Services Market faces challenges such as high operational costs and evolving regulatory requirements. However, ongoing investments in advanced analytical instruments and strategic collaborations are expected to mitigate these challenges.
Looking ahead, the Pharmaceutical Impurity Synthesis and Isolation Services Market is poised for sustained expansion, supported by increasing demand for high-quality pharmaceuticals, regulatory compliance, and continuous innovation. Companies that focus on advanced technologies and customized service offerings will be well-positioned to capitalize on emerging opportunities in the Pharmaceutical Impurity Synthesis and Isolation Services Market.
Q1. What role do impurity synthesis and isolation services play in drug development?
Impurity synthesis and isolation services are critical for identifying, characterizing, and quantifying impurities that may arise during drug manufacturing or storage. These services ensure compliance with regulatory standards and help maintain drug safety, quality, and efficacy throughout the product lifecycle.
Q2. How do global regulatory frameworks influence market demand?
Regulatory agencies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) mandate strict impurity limits and detailed reporting. This drives demand for specialized analytical services, as pharmaceutical companies must meet stringent compliance requirements to gain and maintain market approval.
Q3. What are the most commonly analyzed types of pharmaceutical impurities?
The most common types include process-related impurities, degradation products, residual solvents, elemental impurities, and drug substance or drug product-related impurities. Each type requires specific analytical techniques for accurate detection and characterization.
Q4. How is the rise of biologics impacting impurity analysis services?
Biologics, including monoclonal antibodies and peptides, are structurally complex and sensitive to minor variations. This increases the need for advanced impurity profiling techniques and specialized expertise, creating new opportunities for service providers in the biologics segment.
Q5. What technological innovations are shaping the future of impurity services?
Technologies such as high-resolution mass spectrometry, automated chromatography systems, and AI-driven data analysis are enhancing detection sensitivity, reducing turnaround time, and improving the overall efficiency of impurity characterization.
Q6. Why is Asia-Pacific becoming a hub for impurity service outsourcing?
Countries like India and China offer cost-effective services, skilled talent pools, and expanding pharmaceutical manufacturing infrastructure. These advantages make the region highly attractive for outsourcing impurity synthesis and analytical services.
Q7. How do CROs and CDMOs differentiate themselves in this competitive market?
They differentiate through end-to-end service offerings, advanced analytical capabilities, regulatory expertise, faster turnaround times, and customized impurity solutions tailored to client needs.
Q8. What is the importance of impurity profiling in generic drug development?
For generic drugs, impurity profiling ensures bioequivalence and therapeutic consistency with the original product. It is a critical requirement for regulatory approval and market entry.
Q9. What challenges do service providers face in this market?
Challenges include evolving regulatory requirements, high investment costs for advanced instrumentation, complex analytical procedures, and the need for highly skilled scientists.
Q10. How will strategic collaborations impact market growth?
Collaborations between pharmaceutical companies and service providers enable knowledge sharing, access to advanced technologies, and faster project execution, ultimately accelerating drug development and market expansion.
Our Latest Publication:
Pharmaceutical Impurity Synthesis And Isolation Services Market (2026 – 2035)
Transcatheter Heart Valve Market Witnessing Rapid Growth Driven by Minimally Invasive Innovations
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.