The global Live Biotherapeutic Products (LBPs) and Microbiome Contract Development and Manufacturing Organization.
(CDMO) market is witnessing strong momentum as advances in microbiome science continue to reshape modern therapeutics. Driven by increasing clinical interest, rising chronic disease prevalence, and growing reliance on outsourcing, the market is projected to experience exceptional growth over the next decade.
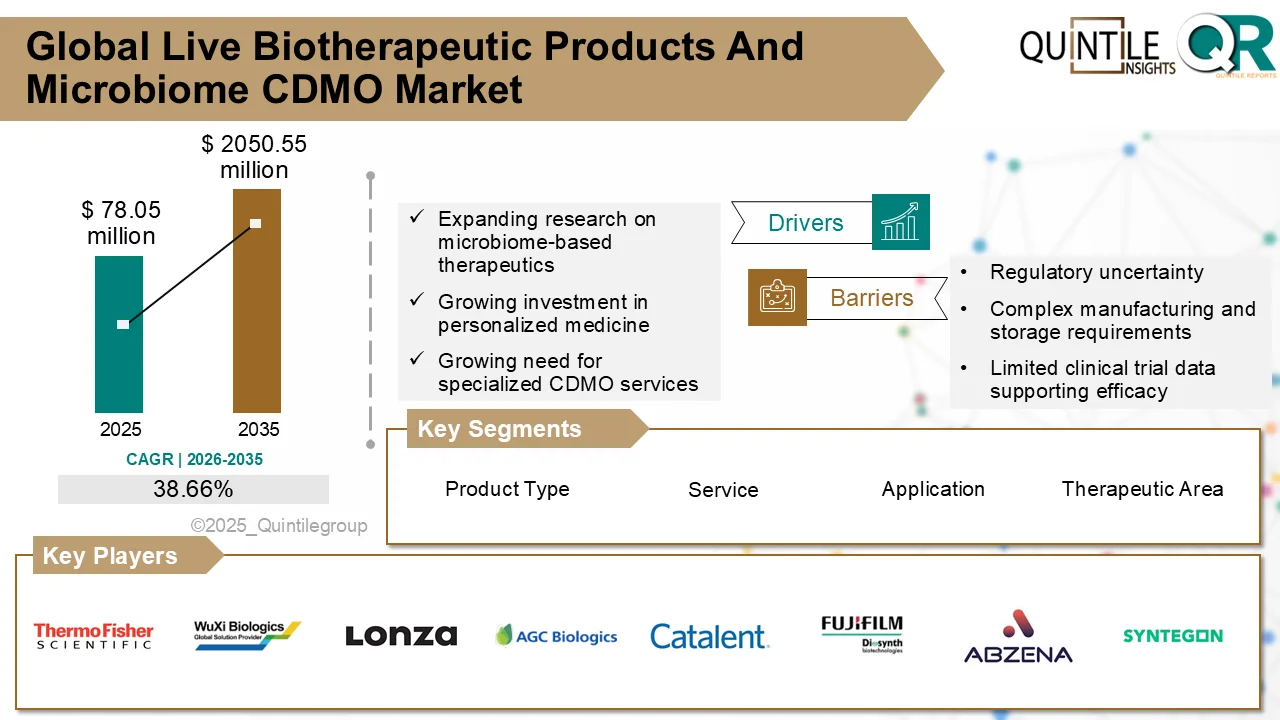
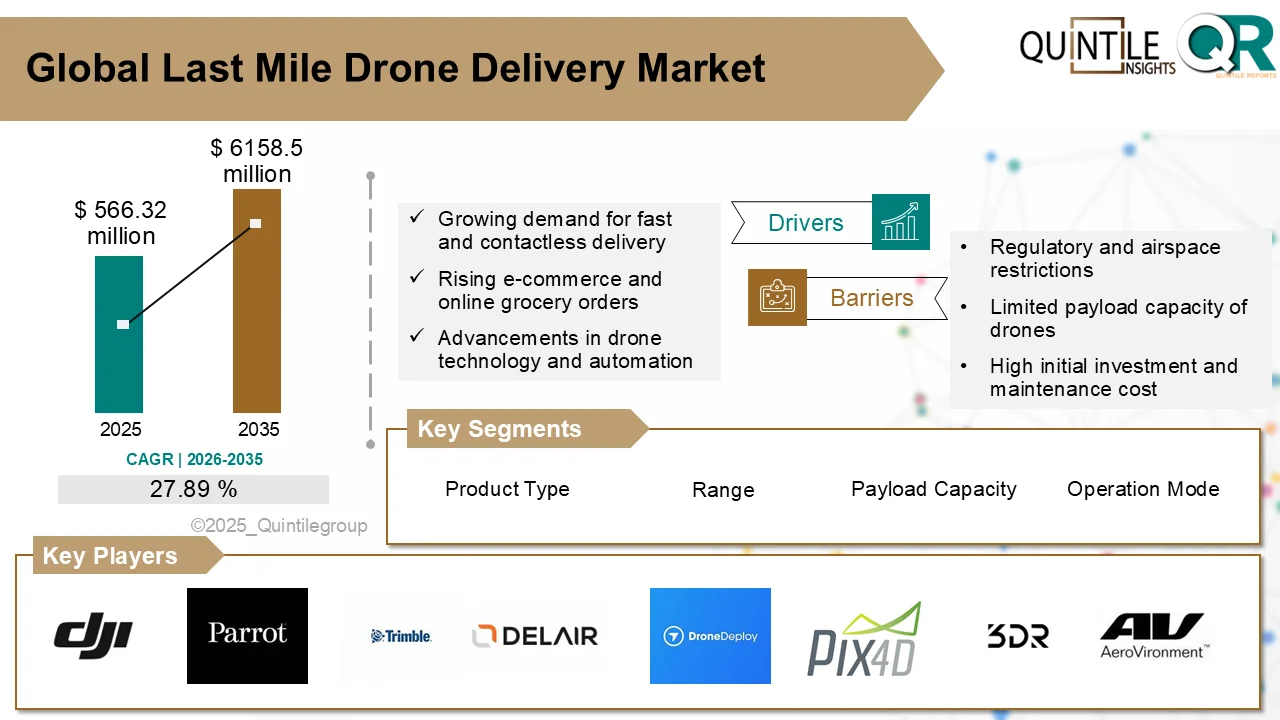
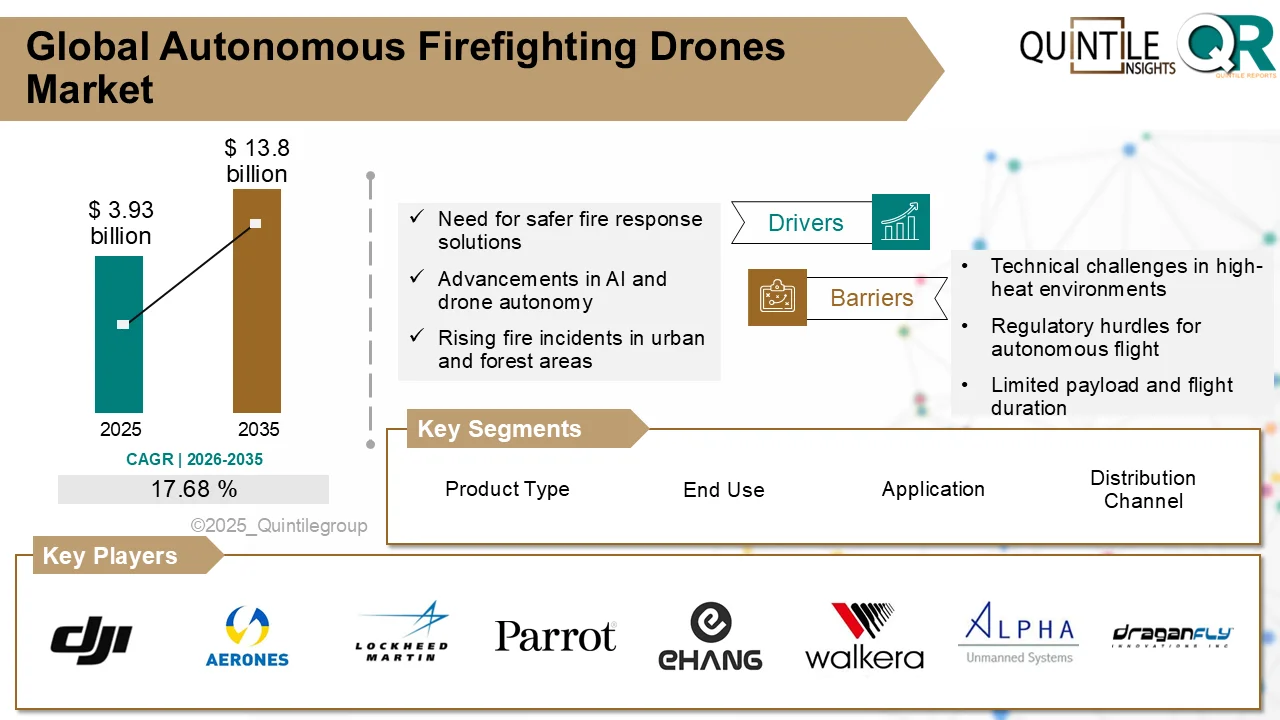
According to industry analysis, the market was valued at USD 78.05 million in 2026 and is forecast to reach USD 2,050.55 million by 2035, expanding at a compound annual growth rate (CAGR) of 38.66% during the forecast period. This rapid rise reflects the growing transition toward microbiome-based therapies and the critical role CDMOs play in enabling their development and commercialization.
Live biotherapeutic products are therapeutic agents composed of live microorganisms designed to prevent, treat, or cure diseases. Unlike conventional probiotics, LBPs are regulated as pharmaceutical drugs and must meet stringent regulatory and manufacturing standards. As a result, pharmaceutical and biotechnology companies increasingly rely on specialized CDMOs to manage complex activities such as strain development, anaerobic fermentation, process optimization, clinical manufacturing, and regulatory compliance.
Request Sample PDF Report: https://www.quintilereports.com/request-sample/1110-live-biotherapeutic-products-and-microbiome-cdmo-market/
One of the primary drivers of market growth is the rising global burden of chronic and lifestyle-related diseases, including inflammatory bowel disease, diabetes, colorectal cancer, obesity, and metabolic disorders. Scientific research continues to highlight the strong link between microbiome imbalance and disease progression, accelerating demand for targeted microbiome-based interventions. With nearly 2 million colorectal cancer cases reported worldwide, therapies targeting gut microbiota are gaining particular attention.
In parallel, pharmaceutical and biotech companies are increasing investments in microbiome drug pipelines. However, the technical complexity of producing live microorganisms—especially oxygen-sensitive strains—has made in-house manufacturing challenging. CDMOs offer the expertise, infrastructure, and quality systems required to scale production while maintaining microbial viability, consistency, and safety.
Place Order: https://www.quintilereports.com/request-enquiry/1110-live-biotherapeutic-products-and-microbiome-cdmo-market/
Regulatory developments are further supporting market expansion. Regulatory agencies in the United States and Europe have introduced clearer guidance for live biotherapeutics, along with expedited pathways such as fast-track and orphan drug designations. These measures are reducing development timelines and encouraging sponsors to advance microbiome candidates into clinical trials.
Despite the optimistic outlook, the market faces several challenges. The number of approved live biotherapeutic products remains limited, with most candidates still in early-stage development. Manufacturing LBPs is highly complex, requiring strict environmental controls and specialized GMP facilities. Additionally, global regulatory standards are still evolving, creating variations in approval requirements across regions. A shortage of skilled professionals with expertise in anaerobic fermentation and microbial quality control also poses a constraint.
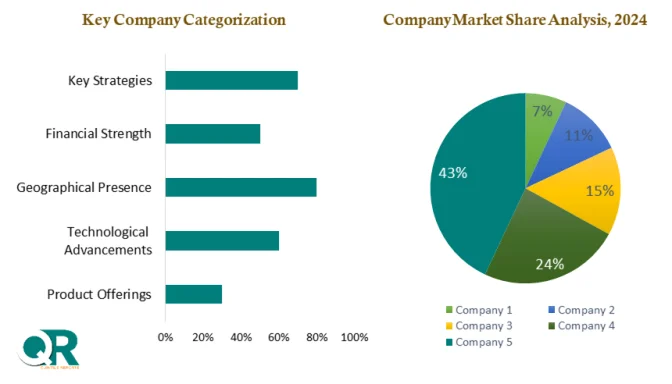
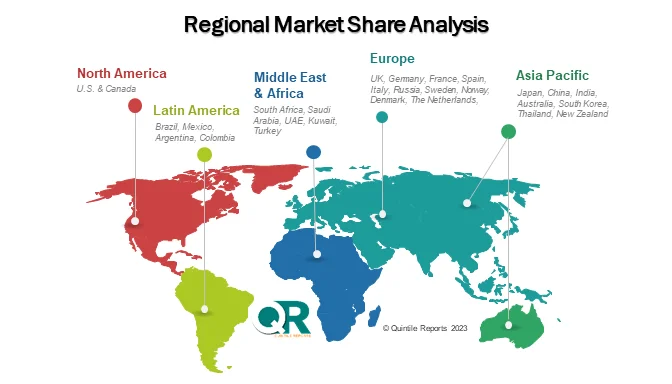
From a regional perspective, North America holds the dominant market position, supported by advanced CDMO infrastructure, a strong microbiome startup ecosystem, and regulatory clarity from the U.S. Food and Drug Administration. Europe follows closely, with countries such as Germany, France, Belgium, and the United Kingdom investing heavily in microbiome research and public-private collaborations. The Asia Pacific region is emerging as a high-growth market, driven by government-backed biotechnology initiatives and expanding CDMO capabilities in countries like Japan, South Korea, China, and India.
The competitive landscape remains relatively concentrated, as only a limited number of CDMOs currently offer comprehensive services for live biotherapeutics. Leading players are expanding capacity through modular GMP facilities and strategic partnerships, while global CDMOs are entering the space through acquisitions and technology upgrades. Key companies operating in the market include Lonza, Catalent, WuXi Biologics, Samsung Biologics, Fujifilm Diosynth Biotechnologies, Thermo Fisher Scientific, AGC Biologics, and Charles River Laboratories, among others.
Looking ahead, continued investment in microbiome research, increasing collaboration between large pharmaceutical companies and biotech startups, and growing demand for precision, non-systemic therapies are expected to sustain strong market growth. CDMOs that combine regulatory expertise, flexible manufacturing platforms, and advanced microbial handling capabilities are likely to play a central role in shaping the future of live biotherapeutic development.
Q1. What are Live Biotherapeutic Products (LBPs)?
Live Biotherapeutic Products are pharmaceutical-grade therapies composed of live microorganisms intended to prevent, treat, or cure diseases. Unlike probiotics, LBPs are regulated as drugs and must meet strict clinical, safety, and manufacturing standards.
Q2. What does the microbiome CDMO market include?
The microbiome CDMO market includes outsourced services such as strain development, process optimization, analytical testing, GMP manufacturing, formulation, fill-finish, stability testing, and regulatory support for microbiome-based therapies and LBPs.
Q3. What is the current and projected market size of the Live Biotherapeutic Products and Microbiome CDMO market?
The market was valued at USD 78.05 million in 2026 and is projected to reach USD 2,050.55 million by 2035, growing at a CAGR of 38.66% during the forecast period.
Q4. What are the key factors driving market growth?
Major growth drivers include rising prevalence of chronic diseases, increasing investment in microbiome-based drug development, growing reliance on outsourcing by pharma and biotech companies, favorable regulatory pathways, and rising demand for targeted therapies with fewer systemic side effects.
Q5. Why are CDMOs critical for live biotherapeutic development?
Manufacturing live microorganisms is technically complex and requires specialized facilities, anaerobic environments, and strict quality controls. CDMOs provide the infrastructure, expertise, and regulatory experience needed to scale production efficiently and safely.
Q6. Which therapeutic areas are driving demand for microbiome-based therapies?
Key therapeutic areas include gastrointestinal disorders, infectious diseases, oncology, immunology, metabolic disorders, neurological disorders, and other chronic conditions where microbiome modulation shows clinical promise.
Q7. What are the major challenges faced by the market?
Challenges include limited commercial approval of LBPs, complex manufacturing requirements, scarcity of GMP-certified facilities, evolving regulatory standards across regions, and a shortage of skilled professionals in microbial manufacturing and quality control.
Q8. Which regions dominate the Live Biotherapeutic Products and Microbiome CDMO market?
North America leads the market due to advanced CDMO infrastructure and strong regulatory clarity. Europe follows with active microbiome research ecosystems, while Asia Pacific is emerging rapidly with growing government support and expanding biomanufacturing capabilities.
Q9. Who are the key players operating in this market?
Prominent companies include Lonza, Catalent, WuXi Biologics, Samsung Biologics, Fujifilm Diosynth Biotechnologies, Thermo Fisher Scientific, AGC Biologics, Charles River Laboratories, Recipharm, BioVectra, and Abzena, among others.
Q10. What is the future outlook for the Live Biotherapeutic Products and Microbiome CDMO market?
The market is expected to witness sustained high growth through 2035, driven by expanding clinical pipelines, increased pharma–biotech collaborations, regulatory clarity, and rising demand for precision microbiome-based therapies.
Our Latest Publication
Live Biotherapeutic Products And Microbiome CDMO Market (2026 – 2035)
Our Latest News:
Non-Alcoholic Wine Market Gains Momentum as Wellness and Innovation Reshape Consumption Habits
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.