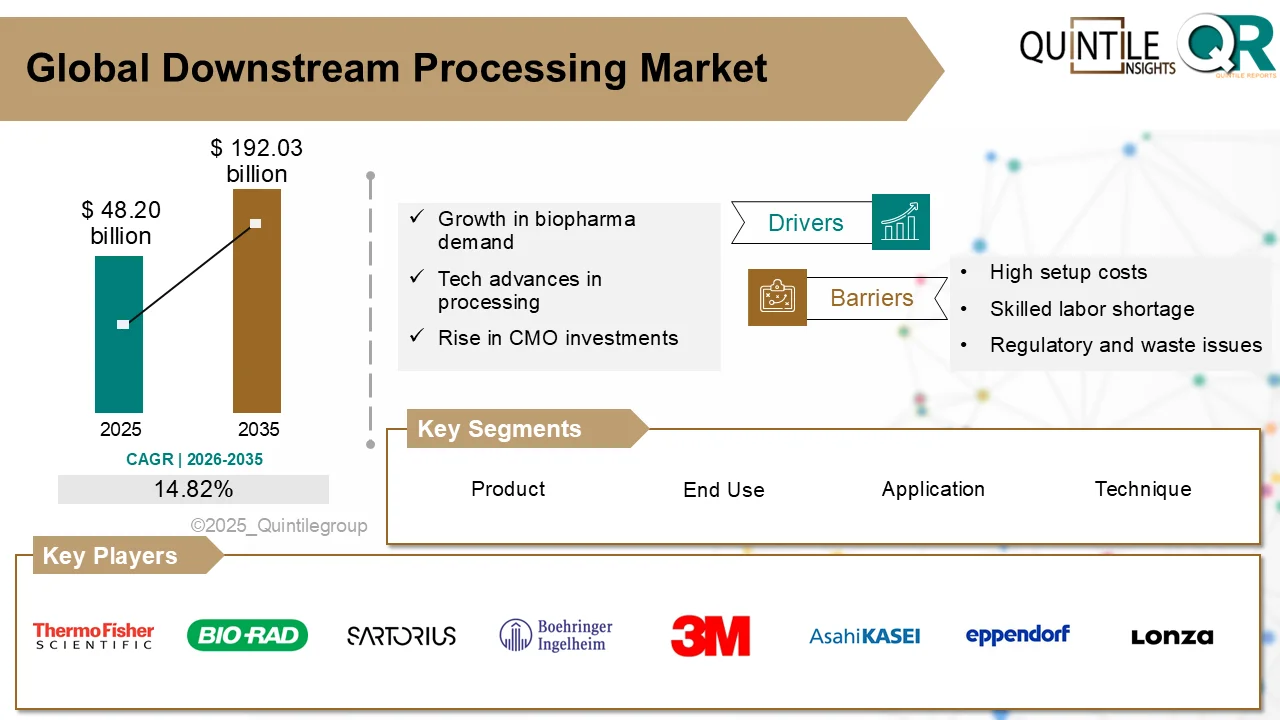
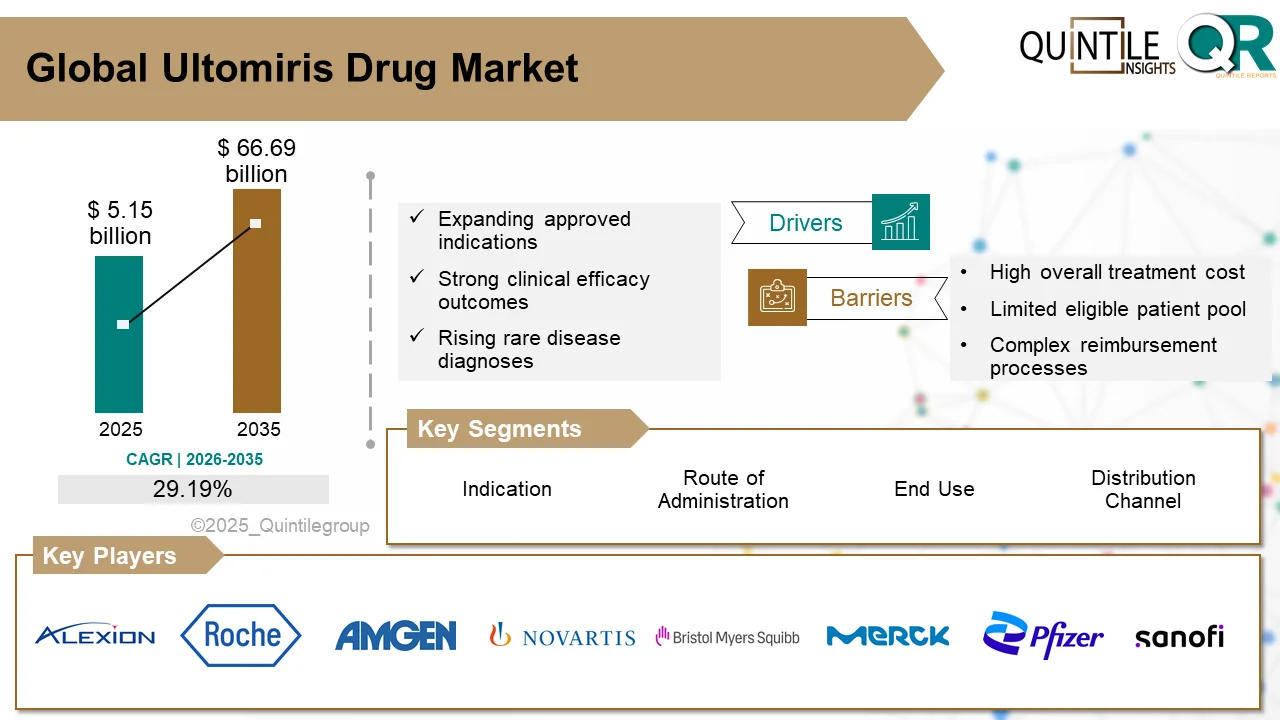
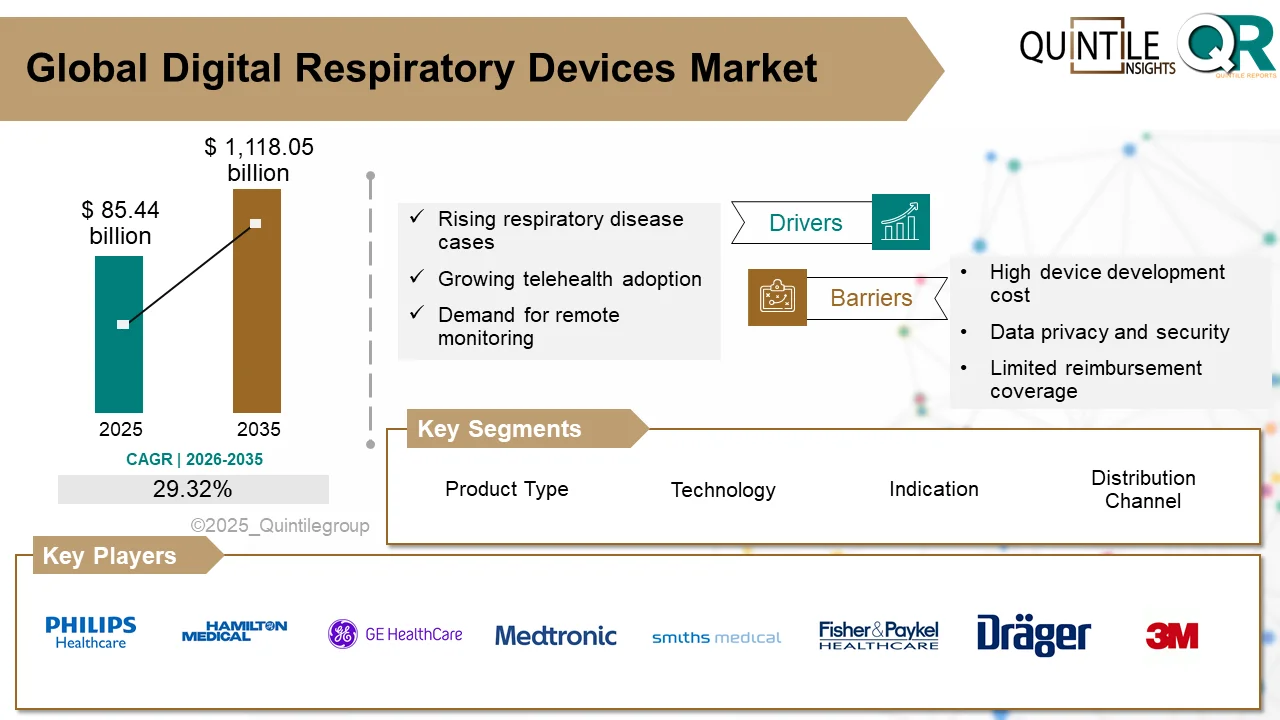
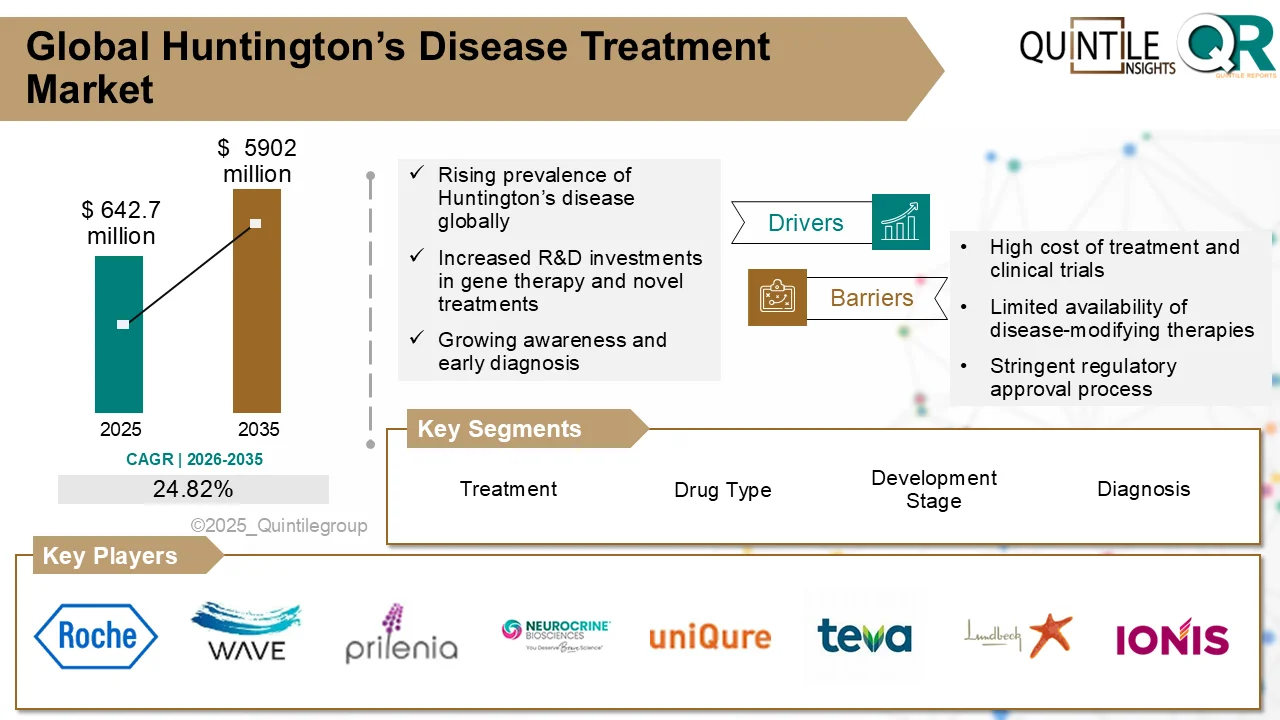
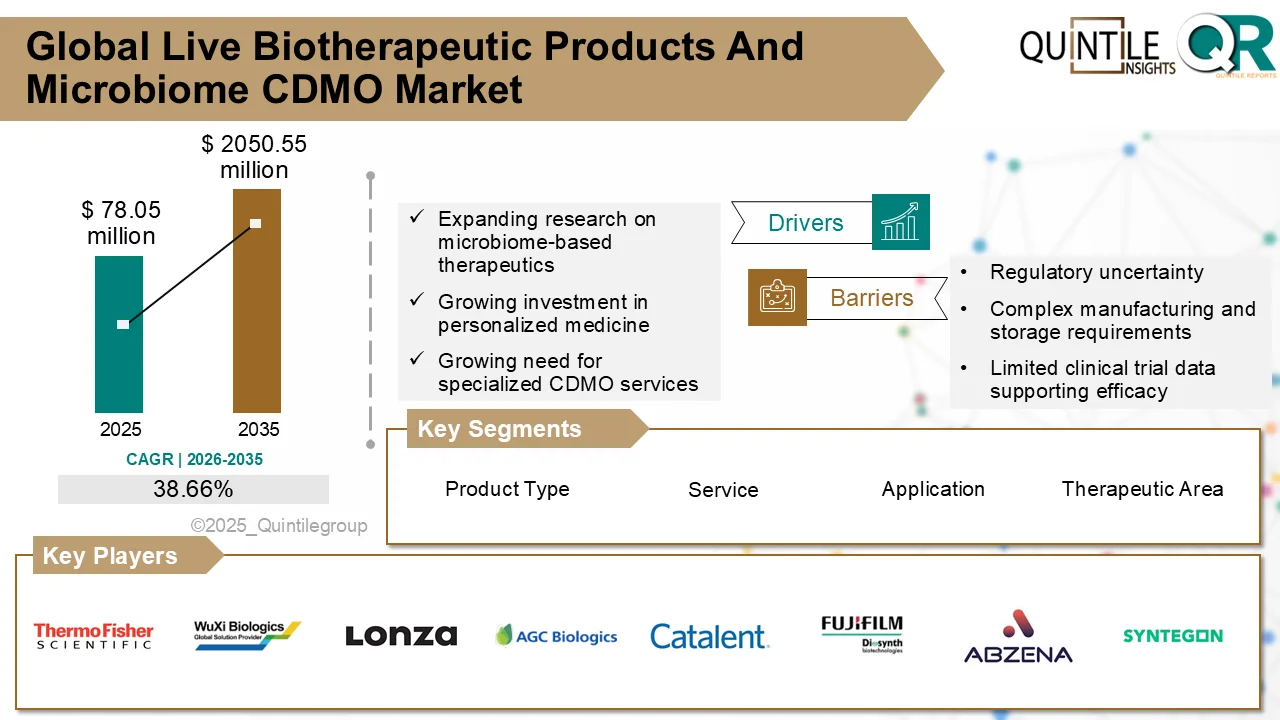
The global Downstream Processing Market is entering a high-growth decade, fueled by accelerating demand for biologics, biosimilars, and advanced therapies.
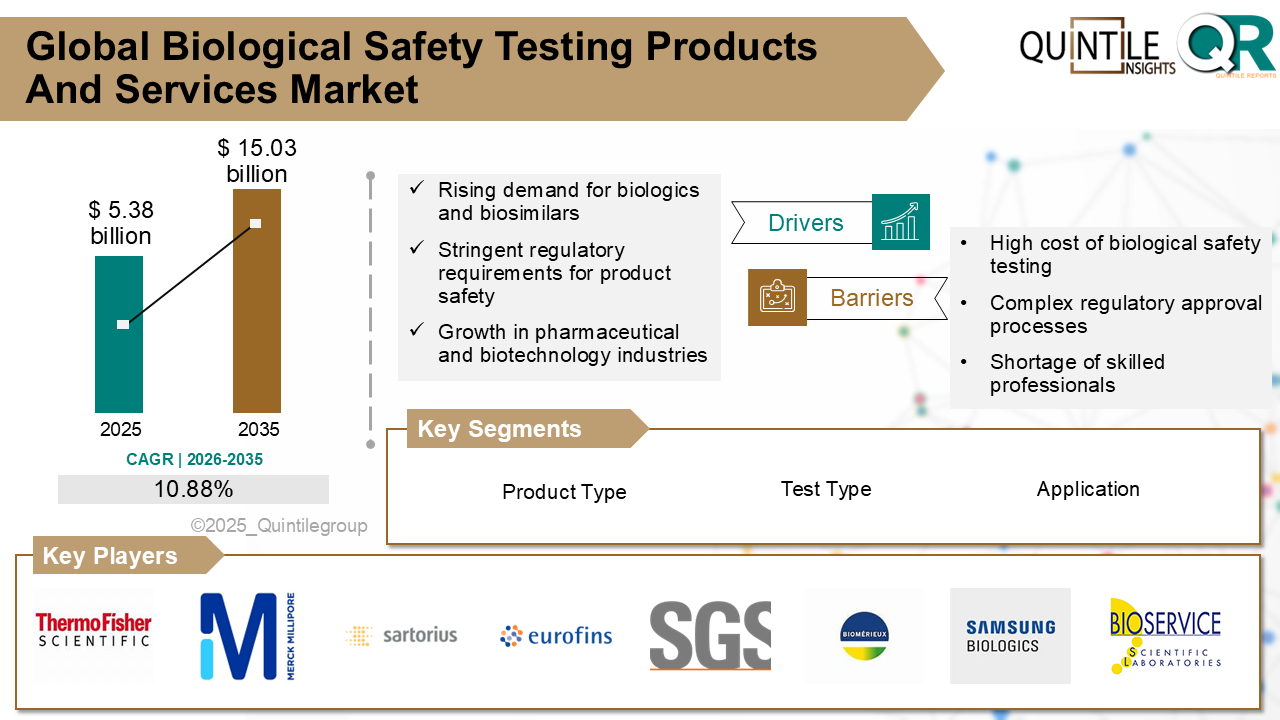
According to analysis by Quintile Reports, the market was valued at USD 48.20 billion in 2026 and is projected to reach USD 192.03 billion by 2035, expanding at a robust CAGR of 14.82% during the forecast period.
Downstream processing represents the critical purification and recovery phase in biopharmaceutical manufacturing, occurring after upstream production such as fermentation or cell culture. It encompasses cell disruption, separation, chromatography, filtration, and polishing steps required to ensure that biologics—including monoclonal antibodies, vaccines, enzymes, and therapeutic proteins—meet stringent regulatory standards for purity, safety, and efficacy.
Request Sample PDF Report: https://www.quintilereports.com/request-sample/1102-downstream-processing-market/
Biologics Expansion Driving Market Momentum
The rapid growth of biologics pipelines is one of the strongest catalysts for downstream innovation. As pharmaceutical and biotechnology companies expand research and development activities, demand for efficient purification systems continues to surge. Increased investment in complex biologics and cell and gene therapies has intensified the need for scalable, high-precision downstream technologies capable of maintaining yield consistency and product integrity.
Simultaneously, outsourcing trends are reshaping the competitive environment. Contract Development and Manufacturing Organizations (CDMOs) are playing a more strategic role in providing integrated downstream services. These firms are adopting modular facility designs and single-use technologies to support flexible, multi-product manufacturing capabilities.
🔥 Limited-Time Discount Available🔥: https://www.quintilereports.com/request-discount/1102-downstream-processing-market/
Technological Advancements Transforming Efficiency
Emerging technologies are redefining the operational landscape of downstream processing. Continuous manufacturing systems, membrane chromatography, tangential flow filtration, and automation tools are enhancing productivity while reducing contamination risks. Integration of Process Analytical Technology (PAT), digital twins, and AI-driven control systems is enabling real-time monitoring and data-driven optimization across purification workflows.
Single-use systems, in particular, are gaining traction for their ability to reduce cleaning validation requirements and operational downtime. Vendors are increasingly emphasizing sustainability and cost reduction by developing energy-efficient equipment and eco-designed consumables aligned with global environmental objectives.
Regulatory Frameworks Shaping Quality Standards
Regulatory compliance remains central to downstream processing operations. In the United States, strict Current Good Manufacturing Practice (CGMP) guidelines govern biologics production, overseen by the U.S. Food and Drug Administration. European manufacturers align with the European Medicines Agency’s rigorous process validation standards, while Japan adheres to comprehensive guidelines set by the Pharmaceuticals and Medical Devices Agency.
These regulatory environments demand meticulous validation of purification steps, viral clearance procedures, and contamination control strategies. Although compliance requirements increase operational complexity, they also drive innovation and quality improvements throughout the industry.
Regional Growth Dynamics
North America continues to lead the downstream processing market, supported by strong biopharmaceutical infrastructure and public-private investment initiatives. Europe maintains a competitive position with advanced engineering capabilities and GMP-compliant production facilities across Germany, the United Kingdom, France, and Switzerland.
Asia-Pacific is emerging as the fastest-growing region, propelled by government-led biomanufacturing programs in China, India, Japan, and South Korea. Expanding biosimilar production and cost-competitive manufacturing environments are strengthening the region’s global footprint. Meanwhile, Latin America and the Middle East & Africa are gradually building purification capacity to reduce reliance on imports and support domestic healthcare priorities.
Buying Now: https://www.quintilereports.com/request-enquiry/1102-downstream-processing-market/
Competitive Landscape and Strategic Investments
The market remains highly competitive, with major players focusing on research expansion, acquisitions, and localized manufacturing strategies. Leading companies such as Thermo Fisher Scientific, Sartorius AG, Merck KGaA, and Danaher Corporation are strengthening end-to-end bioprocess portfolios.
Recent industry developments highlight continued investment in single-use bioreactors, eco-friendly consumables, and expanded manufacturing hubs in Asia. Strategic collaborations with universities and public health organizations are also supporting technology transfer and workforce development.
Outlook Through 2035
Looking ahead, sustained R&D investment, automation adoption, and global regulatory harmonization are expected to drive steady market expansion. Companies that successfully integrate digital technologies, continuous processing, and sustainability initiatives into their downstream platforms will be well positioned to capture long-term growth.
As biologics continue to dominate pharmaceutical pipelines, downstream processing will remain a cornerstone of global biomanufacturing—ensuring that advanced therapies reach patients safely, efficiently, and at commercial scale.
1. What is driving the rapid growth of the downstream processing market?
The primary growth driver is the expanding pipeline of biologics, including monoclonal antibodies, vaccines, cell and gene therapies, and biosimilars. As biologics production increases, the need for advanced purification and recovery systems grows proportionally. Additionally, regulatory pressure for high purity and consistency is pushing manufacturers to adopt innovative downstream technologies.
2. Why is downstream processing considered more complex than upstream processing?
Downstream processing involves purification of large, sensitive biological molecules that vary significantly in structure and stability. Each product requires customized purification workflows involving chromatography, filtration, and viral clearance validation. Yield loss risks and contamination control add further complexity.
3. How are single-use systems impacting the industry?
Single-use technologies reduce cleaning validation requirements, minimize contamination risk, and improve production flexibility. While they introduce recurring consumable costs, they significantly enhance operational efficiency and scalability—particularly for CDMOs and multi-product facilities.
4. What role does automation and AI play in downstream processing?
Automation, AI-driven process control, and digital twins improve monitoring of Critical Quality Attributes (CQAs) and enable real-time adjustments. These tools enhance yield consistency, reduce batch failures, and support regulatory compliance under frameworks such as PAT and Quality by Design (QbD).
5. How do regulatory frameworks influence market growth?
Strict regulatory guidelines in the U.S., Europe, and Japan require comprehensive validation of purification processes. While compliance increases operational costs, it also drives technological innovation and ensures global quality harmonization, supporting long-term market credibility.
6. Which regions are expected to see the fastest growth?
Asia-Pacific is projected to experience the highest growth rate due to government-backed biomanufacturing initiatives in China, India, Japan, and South Korea. North America and Europe remain strong due to established infrastructure and regulatory maturity.
7. How is outsourcing reshaping downstream operations?
Biopharmaceutical companies increasingly outsource purification activities to CDMOs to reduce capital expenditure and accelerate commercialization. This trend is driving demand for modular facilities, flexible purification platforms, and integrated service offerings.
8. What are the key challenges facing manufacturers?
Major challenges include high capital expenditure for chromatography systems, process customization requirements, yield losses during purification, regulatory revalidation burdens, and supply chain disruptions for critical consumables.
9. What competitive strategies are leading players adopting?
Industry leaders such as Thermo Fisher Scientific and Sartorius AG are investing heavily in R&D, acquiring niche purification technology firms, expanding in emerging markets, and promoting sustainable single-use platforms to strengthen competitive positioning.
10. What is the long-term outlook for the downstream processing market?
With a projected CAGR of 14.82% through 2035 and market value expected to approach USD 192 billion, downstream processing will remain a core pillar of global biomanufacturing. Continuous manufacturing, automation, and sustainability integration will define the next generation of purification systems.
Our Latest Publication
Downstream Processing Market (2026 – 2035)
Our Latest News:
Veterinary Dental Equipment Market Poised for Steady Growth Amid Rising Pet Oral Health Awareness (2026–2035)
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.