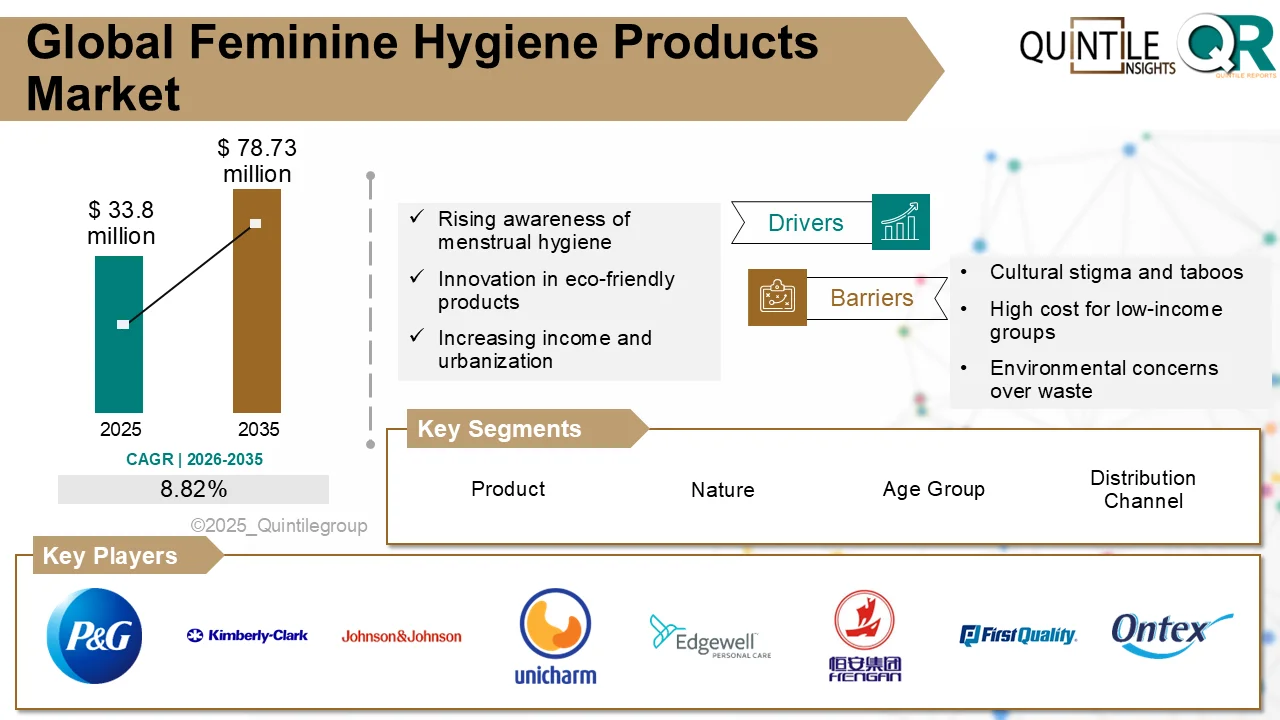
The global Dengue Testing Market is entering a period of sustained expansion, driven by rising infection rates, technological innovation, and enhanced public health surveillance programs.
Valued at USD 637.90 million in 2026, the market is projected to grow to USD 1,234.3 million by 2035, reflecting a steady compound annual growth rate (CAGR) of 6.82% during the forecast period.
Dengue fever, transmitted by Aedes mosquitoes, continues to pose a significant health burden in tropical and subtropical regions. However, shifting climate patterns, increased international travel, and rapid urbanization have expanded the disease’s reach beyond traditional endemic areas. As a result, diagnostic testing has become a critical tool not only for patient management but also for outbreak monitoring and disease control strategies.
The dengue testing ecosystem includes a range of diagnostic solutions such as enzyme-linked immunosorbent assays (ELISA), reverse transcription polymerase chain reaction (RT-PCR) molecular tests, NS1 antigen detection tests, and rapid IgG/IgM antibody kits. These diagnostics are deployed across centralized laboratories, hospitals, diagnostic centers, and increasingly at point-of-care (POC) settings. The growing emphasis on early and accurate detection is fueling demand for high-sensitivity assays that can confirm infection before severe complications develop.
Request Sample PDF Report: https://www.quintilereports.com/request-sample/1107-dengue-testing-market/
Technological progress remains a cornerstone of market growth. Molecular testing platforms are becoming more sophisticated, offering multiplex capabilities that detect dengue alongside other arboviruses such as Zika and chikungunya. Automation in ELISA platforms and portable rapid diagnostic tests are improving turnaround times and expanding access in low-resource environments. These innovations are particularly valuable during outbreak peaks when healthcare systems experience testing surges.
Limited-Time Discount Available: https://www.quintilereports.com/request-discount/1107-dengue-testing-market/
Government initiatives and international health agencies are also playing a vital role in market development. Organizations such as the World Health Organization have intensified efforts to strengthen global preparedness through surveillance programs and response frameworks targeting dengue and other vector-borne diseases. In parallel, the Centers for Disease Control and Prevention has reinforced dengue reporting protocols and recommends NS1 antigen and RT-PCR testing for accurate case confirmation in suspected patients.
Regionally, Asia-Pacific continues to dominate the market due to widespread endemic transmission in countries like India, Indonesia, and Thailand. Strong government-led control programs, expanding laboratory infrastructure, and increasing healthcare spending are supporting both centralized and decentralized testing solutions.
Latin America is emerging as the fastest-growing regional market, fueled by record outbreak numbers and enhanced public health funding in countries such as Brazil and Argentina. Expanded distribution of rapid diagnostic kits and investments in laboratory capacity are strengthening regional preparedness.
In North America and Europe, dengue testing demand is largely driven by travel-related infections and localized outbreaks linked to warmer temperatures. Countries including the United States and Germany are integrating dengue diagnostics into routine screening protocols for febrile travelers returning from endemic areas.
Despite its positive outlook, the market faces certain constraints. Advanced molecular and ELISA-based platforms require substantial capital investment, which may limit adoption in low-income regions. Additionally, variability in regulatory approvals and occasional limitations in rapid test sensitivity can affect market penetration. Seasonal demand fluctuations during monsoon-related outbreaks also create supply chain challenges for manufacturers.
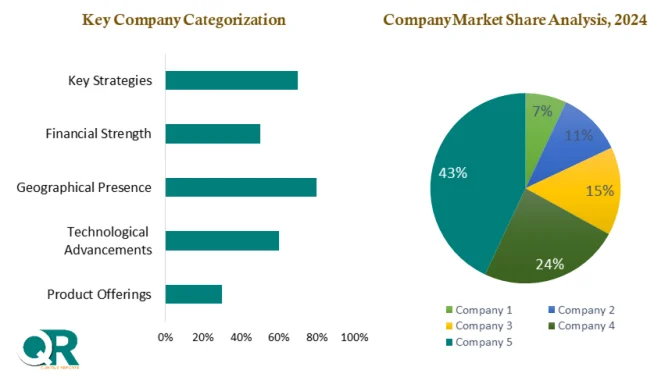
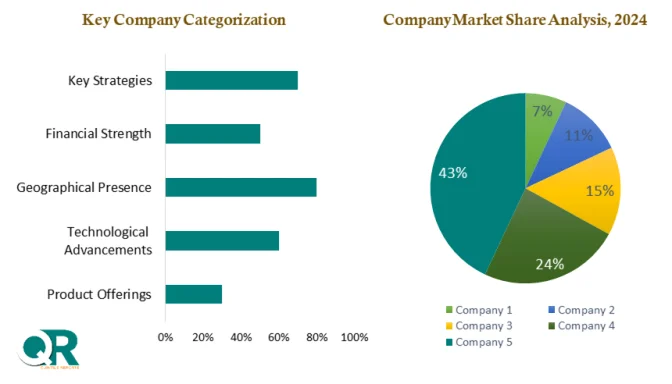
Competition within the market remains strong, with major players such as Abbott Laboratories, F. Hoffmann-La Roche Ltd, Thermo Fisher Scientific Inc., and Siemens Healthineers expanding their diagnostic portfolios. Companies are focusing on improving test accuracy, reducing turnaround time, and developing cost-effective rapid kits to address emerging market needs.
Looking ahead to 2035, continued investments in research and development, public-private partnerships, and digital health integration are expected to shape the future of dengue diagnostics. As global health authorities prioritize outbreak preparedness and climate-related disease surveillance, the dengue testing market is positioned to play an increasingly central role in global infectious disease management.
Buy Now to Access Full Market Insights and Strategic Forecasts: https://www.quintilereports.com/request-enquiry/1107-dengue-testing-market/
1. What is driving the projected growth of the dengue testing market through 2035?
The market is primarily driven by the rising global incidence of dengue infections, expanding mosquito habitats due to climate change, and increased urbanization. Government surveillance initiatives and improved awareness of early diagnosis are accelerating adoption. Additionally, technological advancements in ELISA and RT-PCR platforms are strengthening diagnostic accuracy and scalability.
2. What is the expected market size and growth rate?
The global dengue testing market is projected to grow from USD 637.90 million in 2026 to USD 1,234.3 million by 2035, registering a CAGR of 6.82% during the forecast period.
3. Which diagnostic technologies dominate the market?
ELISA-based assays currently hold the largest market share due to their high sensitivity and reliability in detecting NS1 antigens and IgM/IgG antibodies. However, RT-PCR testing is gaining strong momentum because of its ability to detect dengue in early infection stages and differentiate serotypes. Rapid NS1 antigen and antibody-based point-of-care tests are expanding rapidly in outbreak-prone and low-resource settings.
4. Which regions are leading in market growth?
Asia-Pacific dominates the global market due to high endemicity in countries like India, Indonesia, and Thailand.
Latin America is the fastest-growing region, with strong diagnostic expansion in Brazil and Argentina following record outbreak levels.
In North America and Europe, rising travel-related cases in the United States and Germany are driving demand for screening and surveillance diagnostics.
5. How are public health organizations influencing the market?
Global agencies such as the World Health Organization are implementing preparedness and response strategies to strengthen diagnostic networks worldwide. The Centers for Disease Control and Prevention has also enhanced dengue reporting requirements and recommends NS1 antigen and RT-PCR testing for accurate diagnosis in suspected cases.
6. What are the main challenges limiting market expansion?
Key restraints include:
-
High cost of advanced RT-PCR and ELISA platforms
-
Limited diagnostic infrastructure in rural and endemic regions
-
Variability in regulatory approvals across countries
-
Seasonal fluctuations affecting demand and supply chains
-
Sensitivity limitations in some rapid diagnostic tests
7. Who are the leading companies operating in the dengue testing market?
Major players include:
-
F. Hoffmann-La Roche Ltd
-
Thermo Fisher Scientific Inc.
-
Bio-Rad Laboratories
-
SD Biosensor
These companies are focusing on multiplex testing platforms, automation, and improved point-of-care solutions.
8. How is point-of-care testing shaping the future of dengue diagnostics?
Point-of-care testing is emerging as the fastest-growing segment due to its portability, rapid turnaround time, and suitability for remote and outbreak-prone regions. Integration with mobile health (mHealth) reporting systems is further enhancing real-time surveillance and outbreak management.
9. What opportunities exist for investors and market entrants?
Opportunities include:
-
Development of affordable, high-sensitivity rapid kits
-
Expansion into emerging markets in Asia-Pacific and Latin America
-
AI-enabled diagnostic integration and digital surveillance platforms
-
Strategic partnerships with government health programs
10. What does the market outlook look like beyond 2035?
With increasing emphasis on epidemic preparedness and climate-driven disease monitoring, dengue testing is expected to remain a priority segment within infectious disease diagnostics. Continuous innovation in molecular diagnostics and digital health connectivity will likely define long-term competitive advantage.
Our Latest Publication
Dengue Testing Market (2026 – 2035)
Our Latest News:
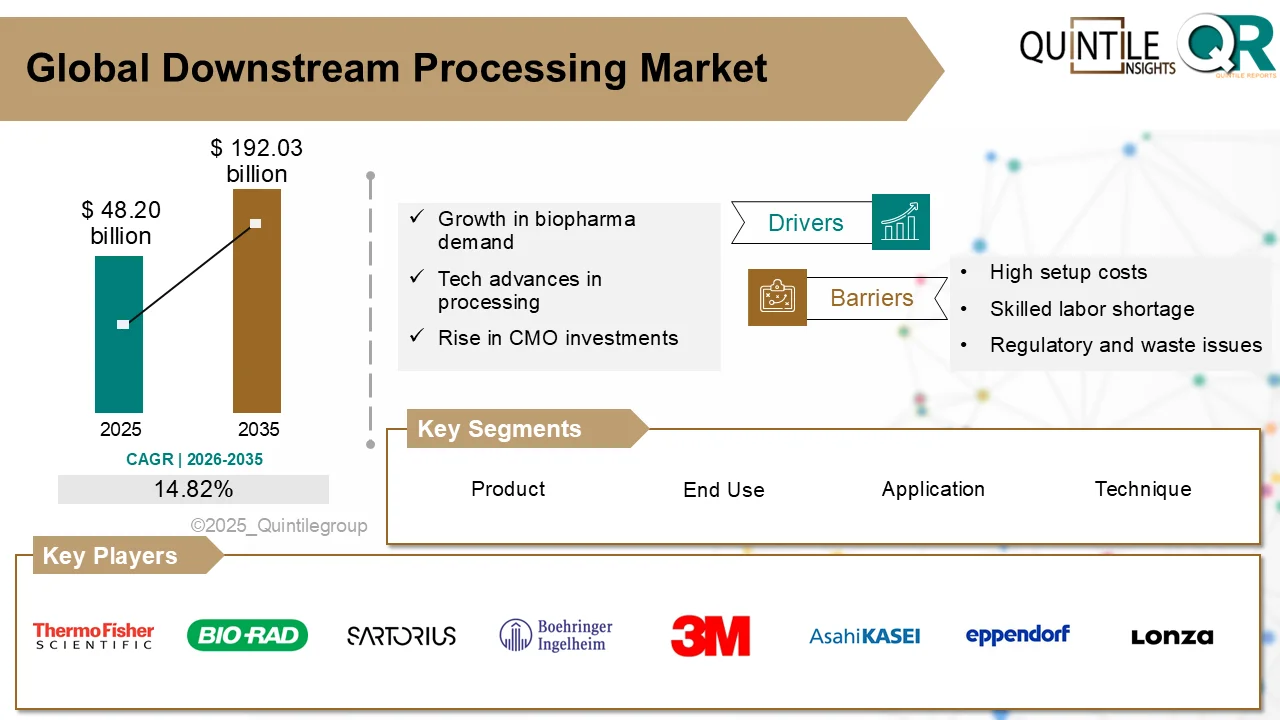
Downstream Processing Market to Quadruple by 2035 Amid Biologics Boom
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.