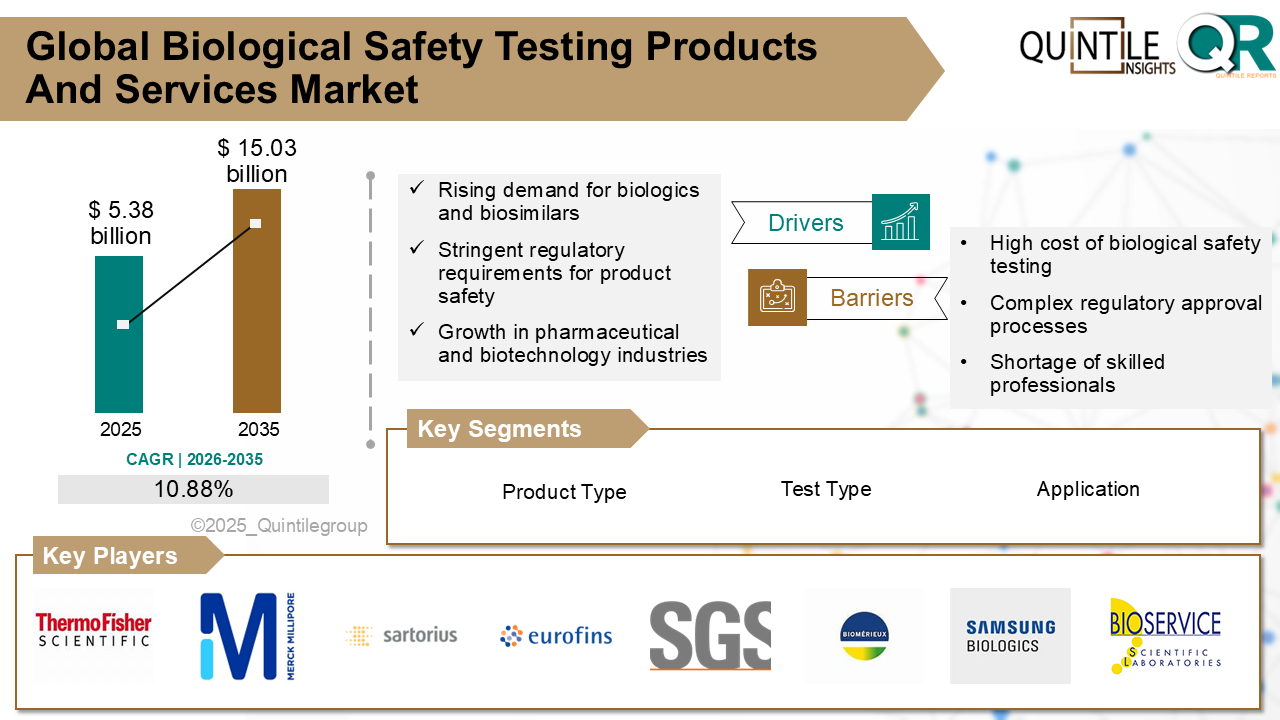
The global biological safety testing products and services market is entering a phase of accelerated expansion, driven by the rapid rise of biologics, tightening regulatory requirements, and continuous.
Technological innovation across the pharmaceutical and biotechnology sectors. According to recent industry analysis, the market was valued at USD 5.38 billion in 2025 and is projected to reach USD 15.03 billion by 2035, registering a robust compound annual growth rate (CAGR) of 10.88% during the forecast period from 2026 to 2035.
Biological safety testing plays a critical role in ensuring the safety, purity, potency, and regulatory compliance of biologics, vaccines, cell and gene therapies, and other advanced therapeutics. As biologics now account for more than 35% of the global pharmaceutical pipeline, safety testing has become a non-negotiable component of modern drug development and commercial manufacturing.
The market encompasses a broad ecosystem of reagents and kits, analytical instruments, and outsourced testing services, covering essential testing areas such as sterility testing, endotoxin testing, viral clearance, adventitious agent detection, bioburden analysis, cell line authentication, and residual host cell contamination testing. These services are delivered by specialized contract research organizations (CROs), contract development and manufacturing organizations (CDMOs), and in-house laboratories within biopharmaceutical companies.
Request Sample Report: https://www.quintilereports.com/request-sample/1214-biological-safety-testing-products-and-services-market/
Key Growth Drivers
One of the most significant drivers of market growth is the intensifying regulatory landscape. Regulatory authorities such as the U.S. Food and Drug Administration (FDA), European Medicines Agency (EMA), and Japan’s PMDA mandate comprehensive, multi-layered biological safety testing for every biologic batch, significantly increasing testing volumes across development and manufacturing stages.
The rapid expansion of cell and gene therapies, growing at an estimated 20%+ CAGR, is further amplifying demand. Each therapy requires extensive viral safety, genetic stability, and sterility testing, increasing both the complexity and value of testing services. Additionally, more than 60% of new drug approvals globally are now biologics, reinforcing long-term demand for safety testing solutions.
Technological advancements are also reshaping the market. The adoption of next-generation sequencing (NGS), high-throughput automation, and AI-driven analytics is reducing testing timelines by up to 30–40%, enabling faster product release while maintaining stringent safety standards. Moreover, the rising financial risk associated with batch failures—where a single contaminated biologics batch can result in losses exceeding USD 550 million—has transformed safety testing from a cost center into a strategic investment.
Place Order: https://www.quintilereports.com/request-enquiry/1214-biological-safety-testing-products-and-services-market/
Challenges and Market Constraints
Despite its strong growth outlook, the market faces several challenges. High testing costs, which can account for 15–25% of total biologics development expenses, create barriers for small and early-stage biotech companies. Additionally, regulatory fragmentation across regions increases compliance complexity and can delay global product launches.
The industry is also grappling with a shortage of skilled biosafety professionals, particularly in advanced molecular and viral testing. In mature markets such as North America and Western Europe, increasing competition has led to margin pressure, making differentiation through technology and regulatory expertise essential.
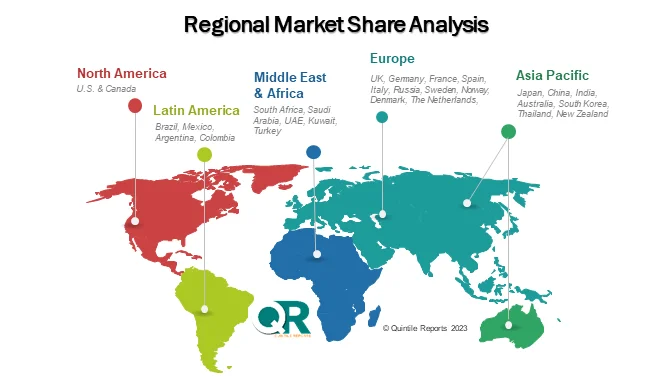
Regional and Competitive Landscape
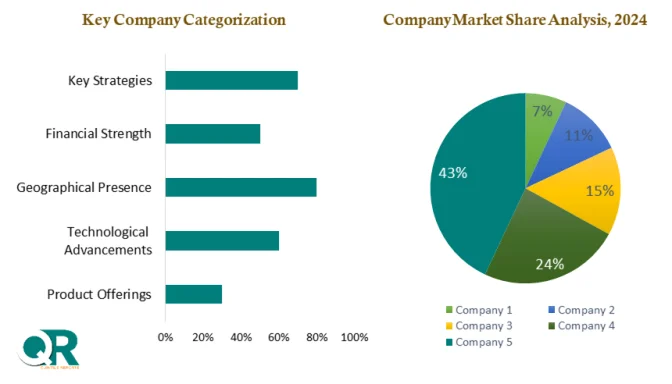
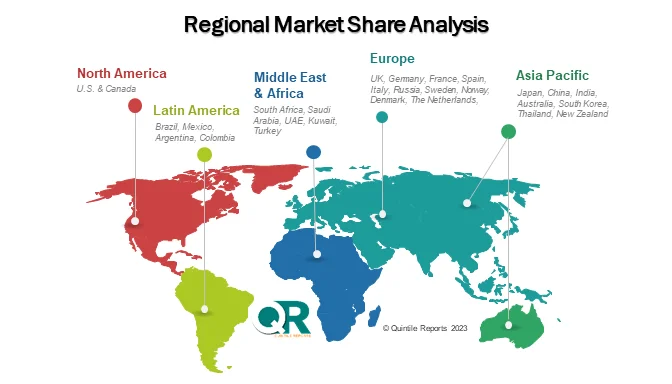
North America dominates the global market, accounting for over 40% of total revenue, supported by strict FDA oversight, heavy R&D investment, and early adoption of automation and AI-enabled testing. Europe follows closely, with Germany and Switzerland leading due to strong pharmaceutical infrastructure and EMA-aligned regulations. Meanwhile, Asia Pacific is the fastest-growing region, expanding at a 10–12% CAGR, driven by scaling biologics manufacturing in China and India.
The competitive landscape is highly expertise-driven, with leading players such as Charles River Laboratories, Thermo Fisher Scientific, Merck KGaA, Sartorius, Eurofins Scientific, SGS, Lonza Group, and WuXi AppTec competing on regulatory credibility, testing breadth, turnaround time, and integration depth. Strategic partnerships, facility expansions, and investment in automation continue to define market leadership.
Outlook
Looking ahead to 2035, the biological safety testing products and services market is expected to sustain its strong growth momentum. Companies that prioritize innovation, regulatory excellence, and scalable testing platforms will be best positioned to capitalize on the expanding global biologics pipeline and the increasing importance of biosafety compliance in modern healthcare.
Q1. What is biological safety testing and why is it important?
Answer: Biological safety testing is a set of analytical procedures used to ensure the safety, purity, potency, and regulatory compliance of biologics, vaccines, cell and gene therapies, and other advanced therapeutics. It is critical because biologics are produced using living systems, making them vulnerable to contamination by viruses, bacteria, or other impurities that could pose serious patient safety risks.
Q2. What is the current market size of biological safety testing products and services?
Answer: The global biological safety testing products and services market was valued at USD 5.38 billion in 2025 and is projected to reach USD 15.03 billion by 2035, growing at a CAGR of 10.88% during the forecast period from 2026 to 2035.
Q3. Which factors are driving the growth of the biological safety testing market?
Answer: Key growth drivers include the rapid expansion of biologics and advanced therapies, increasing regulatory scrutiny from agencies such as the FDA and EMA, rising adoption of next-generation sequencing (NGS) and automation technologies, and the growing financial risks associated with biologics batch failures.
Q4. What types of products and services are included in this market?
Answer: The market includes reagents and kits, analytical instruments, and outsourced testing services. These support critical testing areas such as sterility testing, endotoxin testing, viral clearance, adventitious agent detection, bioburden analysis, and cell line authentication.
Q5. Which applications generate the highest demand for biological safety testing?
Answer: The highest demand comes from vaccines and therapeutics, including monoclonal antibodies and recombinant proteins, followed by gene therapy, blood and blood-based products, tissue-based products, and stem cell therapies.
Q6. What are the major challenges faced by the biological safety testing market?
Answer: Major challenges include high testing costs, regulatory fragmentation across regions, a shortage of skilled biosafety professionals, and intense competition in mature markets such as North America and Western Europe.
Q7. Which region dominates the global biological safety testing market?
Answer: North America dominates the market, accounting for over 40% of global revenue, driven by a strong biopharmaceutical industry, stringent regulatory standards, and significant investments in automation and AI-based testing technologies.
Q8. Which region is expected to grow the fastest during the forecast period?
Answer: The Asia Pacific region is expected to witness the fastest growth, with a CAGR of approximately 10–12%, supported by expanding biologics manufacturing, increasing clinical trials, and improving regulatory frameworks in countries such as China and India.
Q9. Who are the key players in the biological safety testing products and services market?
Answer: Leading players include Charles River Laboratories, Thermo Fisher Scientific Inc., Merck KGaA (MilliporeSigma), Sartorius AG, Eurofins Scientific, SGS SA, Lonza Group, Samsung Biologics, WuXi AppTec, and Promega Corporation, among others.
Q10. What is the future outlook for the biological safety testing market?
Answer: The market is expected to experience sustained growth through 2035, driven by increasing biologics approvals, continuous regulatory tightening, and advancements in testing technologies. Companies that invest in automation, regulatory expertise,
Our Latest Publication
Biological Safety Testing Products and Services Market Size Estimation, Share & Future Growth Trends Analysis, By Product (Reagents & Kits, Instruments, Services), By Application, By Test Type, By and Regional Analysis, 2026-2035
Our Latest News:
Parmesan Cheese Market Set for Steady Growth Through 2035 Amid Rising Demand for Convenience and Premium Dairy
Adarsh
Business Strategy — Quintile Reports
Adarsh is a Business Strategy professional focused on transforming market insights into actionable growth plans. He supports strategic initiatives through market analysis, competitive intelligence, and data-driven decision-making to help drive long-term business success.
His core skills include strategic planning, market research, growth opportunity assessment, trend analysis, performance tracking, stakeholder communication, cross-functional collaboration, and critical problem-solving.